|
P. Kusumakumari,
T.V. Ajithkumar*,
M. Krishnan N air*
From
the Divisions
of Oncology and Radiation
On cology*, Regional Cancer Center, Trivandrum, Kerala, India.
Reprint requests: Dr. P. Kusumakumari, Associate Professor, Division of Pediatric 011cology, Regional Cancer Center, Trivandrum, Kerala 695 011, India.
Manuscript Received: January 5, 1998; Initial review completed: March 5, 1998;
Revision Accepted: May 4, 1998
The common presenting symptoms in childhood acute leukemia are fever, pallor, purpura
and/ or pain. The onset may be insidious or abrupt with any or all of these complaints in varying degrees of severity. A presenting symptom exclusively as soft tissue swelling is uncommon even though subcutaneous nodules are seen in the course of lymphoreticular malignancies. We describe three patients with acute lymphatic leukemia (ALL) whose only symptom
at presentation was soft tissue swelling.
Case Reports
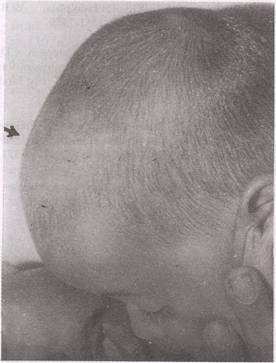
Case 1: A-12-month old first born female baby was first seen in a local hospital with a gradually increasing swelling in the scalp of three months' duration and was referred to the Regional Cancer Center (RCC) with a fine needle aspiration cytology (FNAC) report from the swelling of malignant round cell neoplasm. On physical examination the baby was undernourished and highly irritable.
There was no evidence of any bleeding tendency. A soft non tender swelling of 6 x 4
cm was present over the scalp (Fig. 1). Small discrete lymph nodes were palpable on both sides of the neck. Systemic examination
did not reveal any abnormality. Hematology values revealed hemoglobin 9.5 g/dl, white cell count 7900 cells/cumm and platelet count 63,000 cells/cumm.
Differential count showed 3% abnormal cells. The peripheral smear showed anemia, thrombocytopenia and few blast cells. Bone marrow examination confirmed the diagnosis of ALL (FAB-Ll). Cytogenetic study showed 46 XX t (4,11). Immunophenotyping not done due to lack of facility.
 |
|
Fig. 1. A 12-month-old child with diffuse swelling in the scalp. |
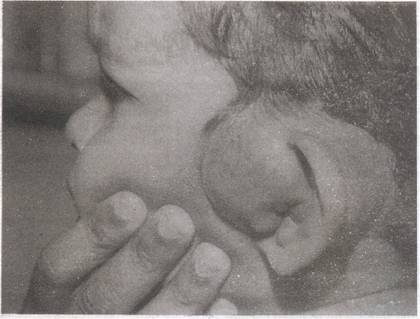
Case 2: A-14-month old male child with swelling involving the left ear lobe of 2 weeks duration had FNAC done in a local hospital which suggested malignant round cell neoplasm. There were no systemic symptoms. Clinical examination at presentation in RCC showed a moderately built and nourished child with no significant lymphadenopathy or organomegaly. The only physical finding was a well defined soft tissue swelling arising from the upper part of left pinna
(Fig.
2). Hemoglobin was 10.5 g/dl, total white cell count 19,200 cells/cumm and platelet count 79,000 cells/ cumm. Differential count showed 31 % abnormal cells. Peripheral smear and bone marrow examination confirmed the diagnosis of ALL (FAB-LI). Cytogenetic study showed 46 XY del 6q21. Other investigations were within normal limits.
Case
3: A 15 month old previously healthy
female baby presented with two months history of swelling over the lateral
aspect of left upper arm. Excision biopsy of the swelling was done in a local
hospital. The child had good general health and there was no evidence of anemia
or bleeding tendency. Systemic examination was unremarkable. Examination of left
upper arm showed a well healed surgical scar on the lateral aspect over the
deltoid insertion with an underlying soft non-tender swelling of 2 x 2 cm. Hemoglobin was 11.9 g/dl, total white cell count 16,800 cells/cumm and platelet count 1,20,000 cells/cumm. Differential count was normal. Peripheral smear did not show any abnormal cells. Biopsy of the tumor and bone marrow examinations were consistent with ALL (FAB-L2). Cytogenetic studies could not be done as no analyzable metaphase cells were identified in the specimen. None of the other investigations revealed any abnormality.
Treatment
Cases 2 and 3 tolerated chemotherapy well and at the time of reporting are into the 8th and 5th month of maintenance chemotherapy,
respectively. There was complete regression of the soft tissue swelling in both. within two weeks of treatment. Case 1 was treated elsewhere and no further information is available.
Discussion
Although ALL is a disease primarily of the bone marrow and peripheral blood, any organ or tissue may be infiltrated by the abnormal cells. Extramedullary
spread is a common feature in ALL and it may either be readily detectable
clinically or demonstrable solely by invasive diagnostic procedures. Although marrow replacement is the major cause of symptoms of leukemia, many important syndromes can result from
extramedullary invasion. The signs and symptoms of a child presenting with ALL reflect the degree of bone marrow infiltration with leukemic cells as well as the extent of extramedullary disease spread. The most common sites of extramedullary spread are central nervous system, testes, liver, kidneys and spleen.
 |
|
Fig. 2. A 14-month-old child with well defined soft tissue swelling arising from the upper part of left pinna. |
Cinically demonstrable soft tissue involvement is uncommon in childhood ALL(l). The three cases reported here showed leukemic deposits in the soft tissues. Except for the soft tissue swelling none of them had lymphadenopathy, visceromegaly or mediastinal adenopathy and all had a total while cell count of less than 20,000 cells/cumm. It is reasonable to presume that soft tissue involvement most likely reflects infiltrationm a metastatic sense(2). However, the prognostic significance of soft tissue swelling at presentation is unknown.
All the three patients were younger than 2 years of age. Most reports state that children. less than 2 years of age at diagnosis, particularly the infants, have a worse prognosis than those diagnosed between the ages 2 and 10 years(3,4).
.
Infants frequently present with features that have been correlated previously with poor out- come in older children with ALL, including hyperleukocytosis, hepatosplenomegaly, central nervous system leukemia, lack of expression of common ALL antigen (CD10) and co-expression of myeloid antigens(5-7). Recent studies have correlated clinical characteristics of infants with unique cytogenetic and molecular anomalies. Approximately 30-50% of infants present with cytogenetically detectable translocation of chromosome
11q23 (most often t[4;11][q21;q23]) (8) and 70-80% have rearrangement of MLLgene, located on the q23 region of chromosome 11 involved in the t(4;11)
translocation, on Southern blot anlayses(9). But this chromosomal abnormality is found only in 2-8% of older children with
ALL(10). In this report one patient had t(4;11) and another had del 6q21, significance of which is not known to us.
Despite advances in therapy for children with ALL, children with age less than 2 years still have a relatively poor prognosis, with most reports showing a long term survival ranging from 20-40%. However, Leiper et al. in their study concluded that patients between 1 to 2 years of age have a prognosis similar to that in older age group(l1). A recent study indicated that event free survival (EFS)
of infants with ALL can be significantly improved with intensification of therapy and they reported a 50 month EFS of 54 +/-11%(7). The two patients in the series who received chemo- therapy attained remission and are on maintenance treatment at the time of reporting.
In conclusion, clinical presentation as soft tissue swelling alone is extremely rare in pediatric ALL. However, It would be worthwhile to bear in mind such a rare presentation.
|
1.
Kennedy BI, Bornstein R, Brunning R.D., Oines D. Breast involvement in acute lymphatic leukemia, Daunorubicin
induced ,remission; Pneumocytis Carinii pneumQnia. Cancer 1970; 25: 693-696.
2.
Nies BA, Bodey GP, Thomas LB, Brecher G, Freirich EJ. The persistence of extramedullary leukemia infiltrates during bone marrow remission of acute leukemia. Blood 1964; 26: 133.
3.. Cangir A, George S, Sallivan M. Unfavorable prognosis of acute leukemia in
infants. Cancer 1975; 36: 1973-1978.
4.
George SL, Fernbach DJ, Vietti H, Sulliwan MP, Lane DM, Haggard ME, et al. Factors influencing survival in paediatric leukemia: The SWCCSG experience Cancer 1973; 32: 1542-1553.
5.
Chessells JM, Eden OB, Bailey CC, Lillyman JS, Richardson SM. Acute lymphoblastic leukemia in infancy: Experience in MRC UK ALL trials: Report from the Medical Research Council Working Party on Childhood Leukemia. Leukemia 1994;8: 1275-1279.
6.
Ferster A, Bertrand Y, Benoit Y, Boilletot A, Beharc, Marguerittle G, et al. Improved survival for acute lymphoblastic leukemia in infancy: The experience of EORTC- Childhood Leukemia Cooperative Group. Br
J
Haematol1994; 86: 284-290.'
7.
Silverman LB, Mclean TW, Gelber RD, Donnelly MI, Gilliland DG, Tarbell NJ, et al. Intensified therapy for infants with acute lymphoblastic leukemia: Results
.
from the Dana-Farber Cancer Institute Consortium. Cancer 1997; 80: 2285-2295.
8.
Heteema NA, Arthur DC, Sather H Albo V, Feusner J, Lange BI, et al. Cytogenetic features of infants less than 12 months of age at diagnosis of acute lymphoblastic leukemia: Impact of the 11 q23 breakpoint on outcome: a report of the Children's Cancer group. Blood 1994; 83: 2274-2284.
9.
Pui C-H, Behm FG, Downing JR, Hancock MC, Schurfleff SA, Ribiero RC, et al. 11 q23/MLL rearrangement confers a poor prognosis in infants with acute lymphoblastic leukemia.
J
Clin Oncol 1994; 12: 909-915.
10.
Arthur DC, Bloomfield CD, Lindquist LL, Nesbit ME. Translocation 4;11 in acute lymphoblastic leukemia: Clinical characteristics and prognostic significance. Blood 1982; 59: 96-99.
11.
Leiper AD, ChesseIs J. Acute lymphoblastic leukaemia under 2 years. Arch Dis Child 1986; 61: 1007-1012.
|
