|
|
|
Indian Pediatr Suppl 2009;46: S20-S26 |
 |
Effect of Child Development Centre Model Early
Stimulation Among At-risk Babies – A Randomized Controlled
Trial |
|
MKC Nair, Elsie Philip, L Jeyaseelan, Babu George, Suja
Mathews and K Padma
From Child Development Centre, Medical College,
Thiruvananthapuram 695 011, Kerala, India.
Correspondence to: Dr MKC Nair, Professor of Pediatrics
and Clinical Epidemiology, and Director,
Child Development Centre, Medical College, Thiruvananthapuram 695 011,
Kerala, India.
E-mail: [email protected]
|
Abstract
Objective: To study the effectiveness of Child
Development Centre (CDC) model early stimulation therapy done in the
first year of postnatal life, in improving the developmental outcome of
at-risk neonates at one and two years of age.
Design: Randomized controlled trial.
Setting and subjects: The study participants
included a consecutive sample of 800 babies discharged alive from the
level II nursery of Medical College, Thiruvananthapuram.
Intervention: The control group received routine
postnatal check-up as per hospital practice. Intervention group in
addition received CDC model early stimulation therapy (home-based).
Results: The intervention group of babies had a
statistically significant higher score for mental developmental index
(MDI) and psychomotor developmental index (PDI) at one and two years of
age. After adjusting all significant risk factors for development, the
babies who had intervention had significantly higher Bayley scores, 5.8
units at one year and 2.8 units at two year, as compared to control
babies.
Conclusion: Early stimulation therapy was
effective at one year. The beneficial effect also persisted at two
years, without any additional interventions in the second year.
Key words: At-risk neonates, Child development, Early
stimulation, Intervention, MDI, PDI.
|
|
T
here is an increasing awareness
among pediatricians on the role of the environment in mental and cognitive
development. Early stimulation programs were developed that targeted
preservation of the mother-infant relationship, improved stimulation for
preterm infants and reduced stress in the neonatal nursery(1). By early
"infant stimulation" we mean early interventional therapy for babies
at-risk for developmental delay. Developmental deficits occur among babies
with genetic and metabolic disorders, environmental risk factors and
biological risk factors like low birth weight(2). Infants born low
birthweight (<1800g) to disadvantaged mothers are at developmental risk
for both biological and social reasons(3).
Meta-analysis of early intervention efficacy studies
has shown that early intervention is effective in improving the
developmental status, although there is no uniform agreement as to whether
the effects last long(4). A recent Cochrane review of sixteen studies has
shown that early intervention programs for preterm infants have a positive
influence on cognitive outcomes in the short to medium-term(5). Long-term
developmental follow-up of at-risk babies in the community, supported by
early intervention therapy needs to be established, as shown by the
experience in the developed countries(6). Large community early
stimulation programs have shown that efficacy was greatest with programs
involving both the parents and the baby; long-term stimulation improved
cognitive outcomes and child-parent interactions, cognition showed greater
improvements than motor skills and, larger benefits were obtained in
families that combined several risk factors(7).
In India, it has been shown that early intervention
program can be successfully conducted through the high risk clinic
approach(8). But, before a national policy is evolved in this regard, a
randomized controlled trial showing efficacy of early inter-vention is
mandatory. Hence this randomized controlled trial was conducted to study
the effective-ness of Child Development Centre (CDC) model early
stimulation therapy, done in the first year of postnatal life, in
improving the developmental outcome among at-risk babies.
Methods
The study was conducted at the level II neonatal
nursery of Sree Avitam Thirunal (SAT) hospital and follow-up was done at
CDC, Medical College, Thiruvananthapuram (Fig. 1). The entry
criteria included; born in SAT hospital, admitted to level II neonatal
nursery, discharged alive and, informed consent for follow-up and early
stimulation. No exclusion criteria were used, so as to give
generalisability to the results obtained. A pilot study on a sample of 100
babies was done to make sure that the randomization procedure, early
stimulation, outcome measure-ments and blinding proceeded as planned.
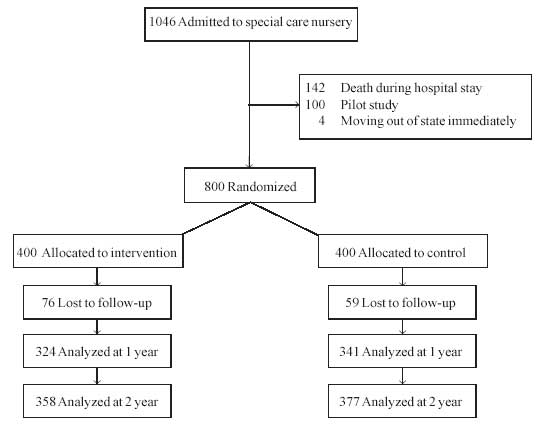 |
|
Fig. 1 Participant flow in the study. |
Sample Size: As this was designed as a
pragmatic clinical trial, the study participants included a consecutive
sample of 800 babies discharged alive, with no exclusion criteria. A
sample size of 336 in each group was obtained, which was adequate to
detect between groups, 4 clinically significant mental developmental index
(MDI), using Bayley scales of infant development (BSID) with alpha error
of 5% and beta error of 20%. A difference of 4 in the MDI scores was taken
as clinically significant difference because it is same as ¼ standard
deviation on Bayley scales, after normalisation of the raw scores to
scores with a mean of 100 and standard deviation of 16 as explained in the
Bayley manual(9,10). Allowing for 20% loss to follow-up, a total of 800
eligible subjects, who met all the inclusion and exclusion criteria were
randomised to intervention and control group on the day of discharge.
Randomization and allocation: Simple
randomisation was done using RALLOC software. Serially numbered opaque
envelops containing the allocation details of a subject were developed at
the Department of Biostatistics, Christian Medical College, Vellore. The
control group received the routine postnatal check-up as per hospital
practice and the intervention group, in addition, received CDC model early
stimulation therapy(11). CDC model early stimulation aims at stimulating
the child through the normal developmental channel, prevention of
developmental delay, prevention of asymmetries and abnormalities,
detection of transient tone abnormalities and minimization of persistent
tone abnormalities. The four major sensory modalities used are; visual
stimulation, auditory stimulation, tactile stimulation and
vestibular-kinaesthetic stimulation. An occupational therapist at CDC,
trained the mothers individually and in groups, to give CDC model early
stimulation and the mothers continued to do the same at home. The
compliance was assessed during monthly follow-up visits by observing the
ease with which the mother did the early stimulation. Compliance score was
derived from a structured questionnaire designed for this purpose, with a
total score of 35, any score below 31 was taken as poor compliance and
above 31 as good compliance.
Outcome measurements were made at one and two years of
age by an observer blind to the treatment status of the babies. These
included MDI, PDI and Bayley score derived as per the Bayley manual. The
Bayley scores represent the motor and mental raw scores together and, MDI
and PDI represent the deviation quotients for mental and motor scores,
respectively. Anthropometric measurements of weight, length and head
circumference were measured as per standard procedure. Those who did not
report for one-year assessment were contacted individually at home before
the second-year assessment.
Quality check of the data collected was done by perusal
of individual data sheets and random checking of about 10% of the
admission record sheets by the principal investigator. Data were entered
and analysed using Foxplus, and SPSS PC+ softwares. For continuous
outcome, Student’s t-test was used to compare the means in the two
groups. Student’s t-test and two-way analysis of variance were used
to compare the means of the study variables between intervention and
control group. A 95% confidence interval for the true difference in means
was also calculated. For multivariate analysis, the study variables, which
were significant at 10% level of significance in bivariate analyses, were
considered. Stepwise multiple regression analysis was done separately for
MDI, PDI and Bayley scores at the end of first and second year.
Significance of the regression model was obtained by F test. R 2
was also computed. Wald test was used to identify the significance of the
variables included in the model. The ethical committee of Medical College,
Trivandrum provided the ethical clearance for the study.
Results
A total of 1046 babies, born in SAT hospital were
admitted to the level II neonatal nursery during the study period. Out of
these 142 babies died in the nursery, 100 babies were used for the pilot
study and 4 babies planned to move outside the state and hence could not
participate in the study. Outcome measurements were available at the end
of one year for 665 babies excluding 135 lost to follow-up, and at the end
of second year for 735 babies excluding 65 lost to follow-up. Table
I shows that the baseline variables are equally distributed in
both arms. Nearly one third of the mothers studied up to middle school. Of
the children studied, nearly 27% were preterm and 50-55% of the babies
were born with low birth weight. Nearly one fourth of them were SGA.
Table II shows one year and two-year outcomes by intervention
and control groups. There was a statistically significant difference
observed with the intervention group having a higher score for MDI, PDI,
Bayley score and length both at one year and two year.
TABLE I
Study Variables at Baseline
Study variable
|
Intervention
n (%) |
Control
n (%) |
|
Residence: Rural |
298 (74.5) |
324 (81.0) |
|
Religion |
|
|
| Hindu |
304 (76.0) |
278 (69.5) |
| Muslim |
56 (14.0) |
55 (13.8) |
| Christian |
40 (10.0) |
67 (16.8) |
| Male sex |
217 (54.3) |
221 (55.3) |
| Caste |
| SC/ST |
33 (8.4) |
42 (10.6) |
| Others |
360 (91.6) |
356 (89.4) |
| Education of father |
| Middle school |
130 (32.8) |
130 (33.0) |
| High school |
266 (67.2) |
264 (67.0) |
| Education of mother |
| Middle school |
125 (31.6) |
134 (33.9) |
| High school |
271 (68.4) |
261 (66.1) |
| Occupation of father |
| Irregular job |
181 (46.3) |
198 (50.5) |
| Employed |
210 (53.7) |
194 (49.5) |
| Occupation of mother |
| Not employed |
352 (90.7) |
354 (90.8) |
| Employed |
36 (9.3) |
36 (9.2) |
| Families with 1 child |
111 (29.0) |
108 (28.4) |
| Monthly income |
| Low |
266 (68.4) |
272 (69.9) |
| High |
123 (31.6) |
117 (30.1) |
| Nuclear family |
98 (25.6) |
115 (29.8) |
| Joint family |
285 (74.4) |
271 (70.2) |
| Birth order >1 |
174 (44.6) |
161 (41.4) |
| Consanguinity |
39 (9.8) |
37 (9.4) |
| High risk pregnancy |
133 (33.6) |
134 (33.6) |
| Assisted delivery |
169 (42.5) |
178 (44.5) |
| Fetal distress |
70 (17.7) |
70 (17.7) |
| Intrauterine infection |
4 (1.0) |
8 (2.0) |
| Pre-term |
108 (27.1) |
111 (27.8) |
| Birthweight <2500g |
202 (50.8) |
217 (54.3) |
| SGA baby |
94 (23.7) |
97 (24.3) |
| Asphyxia |
| Mild |
86 (28.8) |
106 (26.5) |
| Moderate |
10 (3.3) |
16 (4.0) |
| Normal |
203 (67.9) |
278 (69.5) |
| Neonatal seizures |
17 (4.3) |
20 (5.0) |
| Respiratory problems |
65 (16.3) |
62 (15.5) |
| Meningitis |
3 (0.8) |
2 (0.5) |
| CNS Malformation |
2 (0.5) |
2 (0.5) |
| Chromosomal anomaly |
2 (0.5) |
2 (0.5) |
After converting the raw mental, motor and Bayley
scores at 1 year and 2 years to percentile rankings, using tables in the
Bayley manual, the same was compared between intervention and control
group. Percentile ranking position 1 denotes lowest raw scores group (<3rd
percentile) and rank 5 denotes highest raw score group (>50th percentile).
The proportion of babies with rank 1 was higher in the control groups and
less in the intervention groups for one year and 2 year motor and mental
scores. On the other hand, the proportion of babies with rank 5 was higher
in the intervention groups and less in the control groups for one year and
2 year motor and mental scores. These differences observed were
statistically significant. In the intervention group, the mean SD of 1
year and 2 year MDI, PDI and Bayley scores were compared by the compliance
score derived from a structured questionnaire, below 31 denoting poor
compliance for home intervention program and above 31 denoting good
compliance. It was observed that as the compliance score increased, there
was a statistically significant increase in the MDI, PDI and Bayley
scores, both at 1 and 2 years.
TABLE II
Mean and SD of Outcome at One Year and Two Year
| |
Mean |
SD |
Mean |
SD |
P value |
95% CI |
| |
Intervention
(n=324) |
Control
(n=341) |
|
|
|
One Year |
| MDI |
83.6 |
13.7 |
78.5 |
13.3 |
<0.001 |
3.05, 7.15 |
| PDI |
90.9 |
18.1 |
83.7 |
18.2 |
<0.001 |
4.44, 9.96 |
| Bayley Score |
87.3 |
14.1 |
81.1 |
14.2 |
<0.001 |
4.05, 8.35 |
| Weight (kg) |
8.1 |
1.0 |
7.9 |
1.0 |
<0.001 |
0.17, 0.32 |
| Length (cm) |
72.5 |
2.9 |
72.1 |
3.0 |
0.035 |
2.40, 4.39 |
| Head circumference |
44.4 |
2.3 |
44.3 |
1.7 |
0.580 |
–0.206, 0.406 |
Two Year
|
Intervention
(n=324) |
Control
(n=341) |
|
|
| MDI |
83.1 |
13.9 |
80.3 |
13.4 |
<0.005 |
0.83, 4.77 |
| PDI |
99.9 |
15.6 |
95.8 |
16.6 |
<0.005 |
1.77, 6.43 |
| Bayley Score |
91.5 |
13.0 |
88.0 |
13.7 |
<0.005 |
1.57, 5.43 |
| Weight (kg) |
10.3 |
1.4 |
10.1 |
1.3 |
0.089 |
0.005, 0.39 |
| Length (cm) |
83.5 |
3.6 |
82.4 |
5.2 |
0.002 |
0.45, 1.75 |
| Head circumference |
46.1 |
1.8 |
46.3 |
1.7 |
0.173 |
-0.45, 0.05 |
Multiple regression analysis of the study variables for
the outcome measure of Bayley scores at 1 and 2 year was done separately.
Normal birthweight babies had a significantly higher Bayley score, 5.6
units at one year and 6.2 units at two year, as compared to low
birthweight babies. Babies who did not have neonatal seizures had a
significantly higher Bayley score, 7.9 at one year and 9.9 at two year.
Babies who did not have intrauterine infection had significantly higher
Bayley score, 10.8 units at one year and 11.7 units at two, as compared to
others. After adjusting all these significant risk factors for
development, the babies who had intervention had significantly higher
Bayley scores, 5.8 units at one year and 2.8 units at two year as compared
to control babies. The regression models were statistically significant
both at 1year (R 2 = 15.0%, P<0.0001)
and at 2 years (R2 = 18.7%, P<0.0001). Table III
shows that for an increase of every 500 grams, there is a significant and
consistent increase in mean values of Bayley scores, both at one year and
two year. Similarly, in every birth weight group, the mean values were
higher for the intervention group and these differences were statistically
significant. Similar findings were observed for MDI (P=0.005), PDI
(P=0.001), and length (P=0.002), but not for head circumference (P=0.171)
and weight (P=0.090).
TABLE III
Bayley Scores at the Age of One Year and Two Year
| |
Intervention |
Control |
| |
Mean |
SD |
N |
Mean |
SD |
N |
| Bayley score (1 year)* |
| Below 1500g |
83.8 |
12.0 |
38 |
75.3 |
13.2 |
36 |
| 1501 – 2000g |
83.0 |
16.6 |
65 |
80.5 |
15.0 |
67 |
| 2001 – 2500g |
84.4 |
13.4 |
66 |
76.7 |
13.8 |
71 |
| 2501 – 3000g |
91.2 |
12.9 |
100 |
84.2 |
13.1 |
109 |
| 3001 and above |
91.0 |
12.4 |
54 |
85.0 |
14.0 |
58 |
| Bayley score (2 year) |
| Below 1500g |
89.1 |
13.7 |
43 |
81.9 |
13.6 |
43 |
| 1501 – 2000g |
88.0 |
13.5 |
75 |
86.5 |
14.5 |
82 |
| 2001 – 2500g |
87.9 |
14.1 |
71 |
85.1 |
13.3 |
80 |
| 2501 – 3000g |
94.5 |
10.6 |
109 |
91.9 |
11.9 |
110 |
| 3001 and above |
96.4 |
11.9 |
59 |
91.2 |
14.9 |
61 |
* P value <0.001
Discussion
In spite of a long history of mandatory provision of
early intervention programs for at-risk infants in USA, there are still a
few, who genuinely doubt the usefulness of massive state funding for early
intervention programs(12). The term family-centred early intervention
refers to both a philosophy of care and a set of practices, as both have
been used to guide research, training and service delivery(13). Although,
there is no uniform agreement as to the ideal group of babies who would
benefit maximally from early intervention, the neonatal nursery graduates
would probably form the best captive population for providing early
stimulation.
The sample size estimated was a total of 672 and we
have outcome measurements for 665 babies at one year and 735 babies at two
year. In spite of our best efforts, we were not able to evaluate many
babies who missed their one-year assessment appointment date. Home visits
helped to reduce the dropout rate from 17% at one year to only 8% at two
year. Availability of good objective outcome measurements is crucial for
successful completion of any good trial. Hence the objective
neurodevelopmental outcome measurement of MDI and PDI using
internationally accepted Bayley Scales of Infant Development (BSID), which
has been standardized for the Indian population was appropriately used at
one and two years in this study(10).
The intervention group of babies having a statistically
significant higher score for MDI and PDI at one and two year of age,
suggests that not only early stimulation therapy is effective at one year
but also that effect is present even at two years, without additional
intervention in the second year. The observation that, for increase of
every 500 grams, there is a significant and consistent increase in mean
values of the outcomes at 1 and 2 year and that in every birthweight
group, the mean values are higher for the intervention group, again
suggest that early stimulation is effective across the birth weight
groups. Early intervention programs that go into homes have a greater
chance of reaching high-risk infants, compared with those provided at a
distant centre. Better-educated mothers are more likely to be convinced
about the benefits of such inputs(14). In the Indian context, there is a
potential for introducing home-based early stimulation program through the
Integrated Child Development Services. The data provided shows
conclusively that early intervention is effective and hence the results of
this study may have policy implications.
Acknowledgements
Teena Jacoby, Annie John, Leena ML, Asokan N, Child
Development Centre, Medical College, Thiruvananthapuram.
Contributors: MKC was involved in designing the
study and preparation of the manuscript and will act as guarantor. EP was
involved in quality assurance of data, LJ did the analysis of data. BG,
SM, KP were involved in the data collection.
Funding: Kerala Health Research and Welfare
Society, Government of Kerala, Thiruvananthapuram.
Competing interests: None stated. The findings and
conclusions in this article are those of the authors and do not
necessarily represent the views of the funding agency.
|
What This Study Adds?
• CDC model early stimulation therapy done by the
mother at home is effective in improving the developmental status of
neonatal nursery graduates at 1 year and the effect persists at 2
years without additional intervention. |
References
1. Bonnier C. Evaluation of early stimulation programs
for enhancing brain development. Acta Paediatr 2008; 97: 853-858.
2. Nair MKC. Simplified developmental assessment.
Indian Pediatr 1991; 28: 837-840.
3. Scarr-Salapatek S, Margaret L, Williams ML. The
effects of early stimulation on low-birth-weight infants. Child
Development 1973; 44: 94-101.
4. White K, Casto G. An integrative review of early
intervention efficacy studies with at risk children: Implications for the
handicapped. Analysis and Intervention in Developmental Disabilities 1985;
5: 7 –31.
5. Spittle AJ, Orton J, Doyle LW, Boyd R. Early
developmental intervention programs post hospital discharge to prevent
motor and cognitive impairments in preterm infants. Cochrane Database of
Systematic Reviews 2007; 2: CD005495.
6. Nair MKC. Early interventional therapy. Indian J
Pediatr 1992; 59: 657-659.
7. Bonnier C. Evaluation of early stimulation programs
for enhancing brain development. Acta Paediatr 2008; 97: 853-858.
8. Pandit A, Choudhuri S, Bhave S, Kulkarni S. Early
intervention programme through the high risk clinic-Pune experience.
Indian J Pediatr 1992; 59: 675-680.
9. Bayley N. Bayley Scales of Infant Development
Manual. New York: The Psychological Corporation; 1969.
10. Phatak P. Manual for using Bayley Scales of Infant
Development based on Baroda studies and Baroda norms, India : MS
University of Baroda 1973.
11. Nair MKC. Early stimulation CDC Trivandrum Model.
Indian J Pediatr 1992; 59: 663 667.
12. White KR, Mott SE. Conducting longitudinal research
on the efficacy of early intervention with handicapped children. J Div
Early Childhood 1987; 12: 13-22.
13. Bruder MB. Family-centered early intervention.
Early Childhood Special Education 2000; 20: 105-115.
14. de Souza N, Sardessai V, Joshi K, Joshi V, Hughes M. The
determinants of compliance with an early intervention programme for
high-risk babies in India. Child Care Health Dev 2006; 32: 63-72. |
|
|
 |
|

