|
|
|
Indian Pediatr 2017;54: 742-745 |
 |
Long-term Outcome of Inflammatory Bowel
Disease—Unclassified in Children
|
|
Siba Prosad Paul and Bhupinder Kaur Sandhu
From Bristol Royal Hospital for Children, Upper
Maudlin Street, Bristol, UK.
Correspondence to: Dr Siba Prosad Paul, Bristol Royal
Hospital for Children, Upper Maudlin Street, Bristol BS2 8BJ, UK. Email:
[email protected]
Received: February 2, 2016;
Initial review: March 28, 2016;
Accepted: June 13, 2017.
|
|
Objectives: To document the frequency at diagnosis and evolution
over time of inflammatory bowel disease-unclassified in children.
Methods: Analysis of case records (2004-2011) of patients diagnosed
with inflammatory bowel disease-unclassified following
upper-gastrointestinal endoscopy, ileocolonoscopy and small bowel
imaging. Any subsequent diagnostic reclassification by 2016 was
recorded. Results: 344 children diagnosed as inflammatory bowel
disease: 58% Crohn’s disease, 34.5% ulcerative colitis, and 7.5% (n=26)
inflammatory bowel disease-unclassified. 25/26 inflammatory bowel
disease-unclassified patients were followed for 4.5–11.5 years. 17 of
these patients needed endoscopic re-evaluation leading to changed
diagnosis in ten (Crohn’s disease 7, ulcerative colitis 3).
Conclusion: 7.5% (25/344) of inflammatory bowel disease children had
inflammatory bowel disease-unclassified at diagnosis; 10 (40%) evolved
into Crohn’s disease or ulcerative colitis.
Keywords: Crohn’s Disease, Diagnosis,
Ulcerative colitis.
|
|
|
|
T
he revised Porto criteria for diagnosing
Pediatric inflammatory bowel disease (PIBD) was published in 2014, and
reiterated the need for mandatory upper gastrointestinal endoscopy
(UGIE), ileocolonoscopy and small-bowel imaging (preferably magnetic
resonance enterography (MRE)) for all suspected cases [1]. The term
inflammatory bowel disease unclassified (IBDU) previously termed
‘indeterminate colitis’, is reserved for cases of colitis where
following UGIE and ileocolonoscopy, histological findings are not
sufficient to allow a clear differentiation between Crohn’s disease (CD)
and ulcerative colitis (UC), and small bowel imaging is normal [1,2].
There are no clinical or definitive histological features that are
diagnostic of IBDU. However, certain features are more suggestive of
IBDU than UC or Crohn’s colitis, this is described in the diagnostic
features for a child with untreated colitis phenotype in the revised
Porto criteria, 2014 [1]. Category 2 features are rare in UC (<5%) and
category 3 features are uncommon (<5-10% in UC) while predominance of
category 2 features increases the likelihood of CD [1]. Diagnosis of
IBDU should ideally be made jointly by Pediatric gastroenterologist and
Histopathologist.
The objectives of this study were: (i) to
document the frequency of IBDU within the total number of children
diagnosed with IBD in a regional population; and (ii) to document
any change to this diagnosis in the long-term.
Methods
Data were collected at endoscopy for all children
aged 0-17 years diagnosed as PIBD using the Porto criteria over 7-years
(2004-2011). All patients had small bowel imaging (MRE or barium
meal follow-through), Ileocolonoscopy and UGIE. Biopsies (2 to 4 per
site) were obtained from terminal ileum, cecum, ascending colon,
transverse colon, descending colon, sigmoid colon, rectum, duodenum,
pylorus, stomach and esophagus. Repeat endoscopic assessments were
carried out as per clinical indication on patients with persistent
symptoms despite treatment.
All histological specimens were reported by a single
specialist pediatric histopathologist both at initial diagnosis and
reassessment. Information collected included: age at diagnosis, gender,
ethnicity, histological findings, perinuclear anti-neutrophil
cytoplasmic antibodies (pANCA), inflammatory markers (C-reactive
protein, ESR), albumin, liver function tests, full blood count, and urea
and electrolytes. Children with a diagnosis of IBDU were managed as per
the British Society of Paediatric Gastroenterology, Hepatology and
Nutrition guidelines [3].
In 2016 (follow-up period of 4.5–11.5 years),
clinical notes were examined and data on any change in diagnosis from
IBDU to CD or UC, and the histological basis for this change were
collected.
Results
A total of 344 new PIBD patients were diagnosed
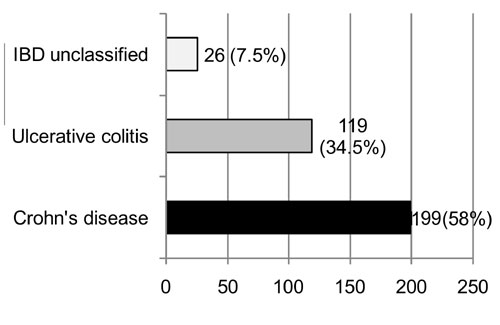
during 2004-2011. Fig. 1 shows the subtypes of PIBD. The
mean age at diagnosis was: 11.5 years for CD (n=199), 11.6 years
for UC (n=119) and 10.1 years (n=26) for IBDU. The age
range of IBDU patients was 1.4 years to 16.1 years; only one child was
aged <2 years in whom cow’s milk protein allergy had been ruled out.
 |
|
Fig. 1 Subtypes of pediatric
inflammatory bowel disease cases over 7 years (n=344).
|
At diagnosis, blood test results were available for
18 IBDU patients with following mean values: platelet count 460 x 10 9/L
(range 142-993), total protein 65 g/L (51- 80), albumin 32 g/L (18-43)
and hemoglobin 11.3 g/dL (8.8-14.1). One patient had family history of
UC. Inflammatory markers (C-reactive protein (CRP) and/or erythrocytic
sedimentation rate (ESR) were abnormal in 12/25 (48%) (6 had raised CRP
and ESR, 3 had rise in either ESR or CRP). The pANCA results were
positive in 11 (44%) patients. It was positive in 4/5 with a revised
diagnosis to CD and 2/3 reclassified to UC. The pANCA results were
missing for 2 children where diagnosis was revised to CD. There were no
significant abnormalities on small bowel imaging at diagnosis in all 25
IBDU cases.
Data were available for 25 out of 26 patients with
IBDU; 16 were males (64%) and 9 females (36%). After a minimum 4.5 years
and maximum 11.5 years follow-up period, case notes of these patients
were examined. Seventeen had been re-evaluated by endoscopic assessment.
Eight were in clinical remission, and had not clinically warranted an
endoscopic reevaluation. Histological features of the group (Group A)
who had revision of diagnosis from IBDU to either CD or UC are
highlighted in Web Table I. Group B consisted of cases
without revision of diagnosis. There were no significant differences at
initial diagnosis in the histology of the eight children who remained in
clinical remission as compared to IBDU patients (n=7) who needed
re-evaluation but had no change in diagnosis.
Table I lists the initial therapeutic
interventions used in 25 IBDU patients. Prednisolone use was similar
between the two groups (P=0.95). Use of aminosalicylates alone
suggestive of milder disease appeared to be higher in Group B but this
was not statistically significant (P=0.49).
TABLE I Therapeutic Interventions Used at Initial Diagnosis
|
Nature of intervention |
Group A |
Group B |
|
(n=10) |
(n=15) |
|
Aminosalicylates alone |
2 |
7 |
|
Exclusive enteral feeding alone |
0 |
1 |
|
Prednisolone alone |
3 |
3 |
|
Prednisolone + Aminosalicylates |
3 |
1 |
|
Prednisolone + Azathioprine |
2 |
2 |
|
Prednisolone + Exclusive enteral feeding |
0 |
1 |
|
Group A: Patients in whom, the diagnosis was revised from
Inflammatory bowel disease- unclassified to Crohn’s disease or
ulcerative colitis; Group B: Patients in whom, the diagnosis of
inflammatory bowel disease-unclassified was not revised on
follow-up |
The median follow-up to revision of diagnosis was 51
months (range 34-87 months). At follow-up (by 2016) diagnosis of IBDU
had changed in 10/25 (40%) cases; 7 to CD and 3 to UC. Two of these 10
patients required hemicolectomy (1 CD, 1 UC). Preoperative assessment
endoscopy was considered inappropriate in these two patients as they
were too sick. Detailed histological results were not available for
three patients (2 transferred to their local hospital’s adult
gastroenterology services and 1 migrated to another region).
Discussion
In this study from a single Pediatric
gastroenterology center strictly following Porto diagnostic criteria,
IBDU comprised only 7.5% (26/344) of total IBD patient at initial
diagnosis. Children with IBDU were younger than those with CD or UC and
there was male preponderance. After a median follow-up period of 52
months, the diagnosis of IBDU needed revision in 10 (40%) children; 7 to
CD and 3 to UC.
The study had some limitations. It included review of
clinical notes in 2016 (4.5-11.5 years after initial diagnosis of IBDU),
and was not a continuous longitudinal study. Some data were missing from
the clinical notes, and thus unavailable for final analysis. Some blood
results were not available as patients referred from secondary care
hospitals had initial investigations done locally. Testing for Anti-Saccharomyces
cerevisiae antibodies was not offered by our laboratory. For three
patients who had moved to other centers, only summaries of change of
diagnosis but not detailed histology following repeat endoscopic
assessment were available
Previous studies have recorded much higher proportion
(12.7-22%) of IBDU out of total PIBD patients [4,5]. The frequency of
IBDU in our series was much lower (7.5%), and is in concordance with
only one recent multicenter European study with 3641 children with IBD
diagnosed using the Porto criteria (EUROKIDS Registry) where IBDU
frequency at initial diagnosis was 7.7% [6]. These low percentages in
our study and EUROKIDS study are likely to be a reflection of strict
adherence to the Porto criteria for diagnosis of PIBD. In our study,
over time, IBDU decreased from 7.5% to 4.3% which is similar to the
EUROKIDS study where IBDU decreased to 5.6% during a median follow-up of
5.7 years [6]. A retrospective US study carried out pre-publication of
the Porto criteria with 78 IBDU children, documented a lower age at
diagnosis [9.2±4 years], and 23% IBDU were reclassified: 8 CD, 5 UC, and
5 non-IBD conditions [7]. A 6-center retrospective study with 210 PIBD
patients, IBDU was reclassified in 8/20 (40%) patients, median time to
revision was 18.5 months [8]. In our study, use of prednisolone and
aminosalicylates at initial diagnosis was similar between those whose
diagnosis changed (Group A) and those whose diagnosis remained unchanged
(Group B). A retrospective multicenter study which pooled data on
roughly equal numbers of patients with CD, UC and IBDU, 260/797 had IBDU
[9]. These IBDU patients had milder disease course, with lower
medication burden and need for surgery [9].
Our study suggests that it is essential to strictly
follow the recommended Porto criteria for optimizing diagnosis of IBD
and specially IBDU. Early repeat reassessment with endoscopy and
small-bowel imaging in cases with persistent symptoms or where surgery
may be a possibility should be considered. As treatment of CD and UC
differs considerably, the treatment for changed correct diagnosis is
essential.
Contributors: SPP: study design, data collection,
analysis, manuscript preparation and revision; BKS: concept,
supervision, manuscript editing and revision, and provided expert
opinion.
Acknowledgements: Dr Christine Spray and
Dr Dharamveer Basude, Consultant Pediatric Gastroenterologists, Bristol
Royal Hospital for Children.
Funding: None. Competing interests: BKS
was a founder member of ESPGHAN working group on IBD (The Porto Group)
and served on the group until 2015.
|
What This Study Adds?
• This study documents a low frequency of
IBDU at diagnosis of PIBD
• Around 40% of IBDU cases can evolve into CD or UC over
long-term where the diagnosis is made as per Porto criteria and
the initial frequency is low.
|
References
1. Levine A, Koletzko S, Turner D, Escher JC,
Cucchiara S, de Ridder L, et al. ESPGHAN revised porto criteria
for the diagnosis of inflammatory bowel disease in children and
adolescents. J Pediatr Gastroenterol Nutr. 2014;58:795-806.
2. IBD Working Group of the European Society for
Paediatric Gastroenterology, Hepatology and Nutrition. Inflammatory
bowel disease in children and adolescents: recommendations for diagnosis
– the Porto criteria. J Pediatr Gastroenterol Nutr. 2005;41:1-7.
3. Sandhu BK, Fell JM, Beattie RM, Mitton SG, Wilson
DC, Jenkins H; IBD Working Group of the British Society of Paediatric
Gastroenterology, Hepatology, and Nutrition. Guidelines for the
Management of Inflammatory Bowel Disease in Children in the United
Kingdom. J Pediatr Gastroenterol Nutr. 2010;50:S1-13.
4. Prenzel F, Uhlig HH. Frequency of indeterminate
colitis in children and adults with IBD - a metaanalysis. J Crohns
Colitis. 2009;3:277-81.
5. Turunen P, Kolho KL, Auvinen A, Iltanen S, Huhtala
H, Ashorn M. Incidence of inflammatory bowel disease in Finnish
children, 1987-2003. Inflamm Bowel Dis. 2006;12:677-83.
6. Winter DA, Karolewska-Bochenek K,
Lazowska-Przeorek I, Lionetti P, Mearin ML, Chong SK, et al.
Pediatric IBD-unclassified is less common than previously reported;
Results of an 8-year audit of the EUROKIDS Registry. Inflamm Bowel Dis.
2015;21:2145-53.
7. Malaty HM, Mehta S, Abraham B, Garnett EA, Ferry
GD. The natural course of inflammatory bowel disease-indeterminate from
childhood to adulthood: within a 25 year period. Clin Exp Gastroenterol.
2013; 23:115-21.
8. Newby EA, Croft NM, Green M, Hassan K, Heuschkel
RB, Jenkins H, et al. Natural history of paediatric inflammatory
bowel diseases over a 5-year follow-up: A retrospective review of data
from the register of paediatric inflammatory bowel diseases. J Pediatr
Gastroenterol Nutr. 2008;46: 539-45.
9. Aloi M, Birimberg-Schwartz L, Buderus S, Hojsak I,
Fell JM, Bronsky J, et al. Treatment options and outcomes of
pediatric IBDU compared with other IBD subtypes: A retrospective
multicenter study from the IBD Porto Group of ESPGHAN. Inflamm Bowel
Dis. 2016;22:1378-83.
10. Birimberg-Schwartz L, Zacker DM, Akriv A,
Cucchiara S, Cameron FL, Wilson DC, et al. Development and
validation of diagnostic criteria for IBD subtypes with an emphasis on
IBD-Unclassified in children: A multicenter study from the Pediatric IBD
Porto group of ESPGHAN. J Crohns Colitis. 2017 Apr 18 [Epub ahead of
print].
|
|
|
 |
|

