|
|
|
Indian Pediatr 2009;46: 883-885 |
 |
Ferrous Sulfate Versus Iron Polymaltose
Complex for Treatment of Iron Deficiency Anemia in Children |
|
Ankur Vikas Bopche, Rashmi Dwivedi, Rakesh Mishra and GS Patel
From the Department of Pediatrics, Kamla Nehru Hospital,
Gandhi Medical College and Associated Hamidia Hospital, Bhopal, MP, India.
Correspondence to: Dr Rakesh Mishra, Department of
Pediatrics, Gandhi Medical College and Associated Hamidia Hospital,
Bhopal, MP, India.
Email: rakeshgastro@yahoo.co.in
Manuscript received: October 19, 2007;
Initial review: March 10, 2008;
Accepted: August 28, 2008.
Published online 2009 April 15. PII:S097475590700648-2
|
|
Abstract
We assessed the clinical response and side effects of
Ferrous sulfate (FS) and Iron polymaltose complex (IPC) in 118 children
with Iron deficiency anemia (IDA). Subjects were randomized to receive
therapy with either oral IPC (Group A, n=59) or oral FS (Group B,
n=59); all were given elemental iron in three divided doses of 6
mg/ kg/day. One hundred and six children could be followed up; 53 in
each group. Children who received ferrous sulfate were having higher
hemoglobin level, and less residual complaints as compared to those who
had received iron polymaltose complex. Our study suggests ferrous
sulfate has a better clinical response and less significant adverse
effects during treatment of IDA in children.
Key words: Ferrous sulfate, Hemoglobin, Iron deficiency
anemia, Iron polymaltose complex.
|
|
A joint UNICEF/USAID consultation has
recommended that the most practical iron supplement for use in infants and
young children should be an aqueous solution of a soluble ferrous salt,
such as ferrous sulfate (FS) or a ferric complex, such iron
polymaltose(IPC)(1). Both of them have been demonstrated to have
equivalent bioavailability in infants(2,3). There is an ongoing debate
over the efficacy of IPC in the background of pressure marketing done by
the manufacturers and lack of data in the Indian context. This study was
thus designed to compare the efficacy and side-effects of IPC versus the
conventional FS preparations in treatment of iron deficiency anemia (IDA).
Methods
This randomized clinical trial was conducted in a
teaching institution with a tertiary level pediatric centre in central
India over a period of one year (October 2004 – September 2005) to
determine the therapeutic efficacy of two different oral iron
preparations: iron polymaltose complex (IPC) and ferrous sulfate (FS). All
the OPD patients of either sex, age ranging from 1-6 years with clinical
features suspected of iron deficiency anemia (IDA) were assessed for
eligibility. In addition, children without clinical manifestations whose
blood had been tested for some other purpose and were found to have Hb <10
g/dL were also included for the study. We assessed 154 young children with
suspected IDA, of which 118 were confirmed to have IDA by serum iron
chemistry. These children were randomized to receive therapy with either
oral IPC (Group-A: Syrup Mumfer ®,
n=59) or oral FS (Group-B; Tablet Nesfol®, n=59) (Fig.1).
All were given elemental iron in three divided doses of 6 mg/kg/day, 30
minutes before meals. Syrup Mumfer® was purchased from the market and
Tablet Nesfol® was freely available from the hospital. Randomization was
achieved by simple randomization and allocation was concealed by sealed
envelope technique. All children were dewormed before start of therapy and
were asked to avoid tea, coffee and phytates. The patients were asked to
return for follow up after 1 month. Compliance and side effects were
checked by verbal enquiry. Verification was done by checking the used
bottles and wrappers of tablets. Resolution of symptoms and signs were
evaluated on follow up and a repeat hemoglobin was done.
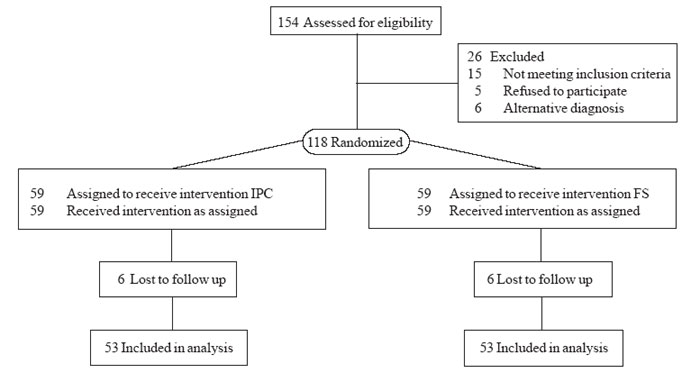 |
|
Fig. 1 Study flow chart. |
Data were recorded on a pre-designed performa,
tabulated and the result were analysed statistically by statistical
package for social sciences (SPSS). Chi square test was applied to
calculate statistical significance. A P value of <0.05 was
considered statistically significant. The maximum permissible type II
error is 20%. Our institutional review committee for ethical research
approved the study. Written informed consent from parents was obtained
prior to enrollment of the subjects in the study.
TABLE I
Therapeutic efficacy of Iron Polymaltose Complex (IPC) and Ferrous Sulfate (FS)
Therapeutic efficacy
|
Group A |
Group B |
|
IPC (n=53) |
(FS) (n=53) |
| Hb at enrolment (g/dL) |
8.46+0.73 |
8.53+0.84 |
| Hb at followup (g/dL) |
8.67+0.73 |
9.44+0.67 |
| P value |
>0.05 |
<0.01 |
| Residual complaints [(n(%)] |
16 (30.8%) |
2 (3.8%) |
| Side effect [(n(%)] |
4 (7.6%) |
9 (17.0%) |
| Increase in Hb [(n(%)] |
38 (71.7%) |
52 (98.1%) |
|
Hb: Hemoglobin. |
Results
Table I provides the outcome measures in the
two groups. Majority of cases in both Group A and B showed rise in
hemoglobin after treatment. No change in Hb was observed in 7.6% (n=4)
children in group A and 1.9 % (n=1) in Group B. Eleven (20.75%)
cases had decrease in the hemoglobin in Group A while no case showed
decrease in hemoglobin in Group B.
Gastrointestinal side effects were 2.5 times more
common in FS group as compared to IPC group (Odds ratio=0.4; 95% CI:
0.35-0.45). As a whole residual complaints were more common in IPC group
as compared to FS group at one month follow up (Odds ratio=11.1; 95% CI
11.04-11.15). The cost of 100 mg elemental iron in form of IPC was four
rupees against two rupees for equal amount of elemental iron from ferrous
sulfate.
Discussion
We conducted this study to compare therapeutic efficacy
of FS versus IPC in the treatment of IDA in children. This study shows
that number of children showing increase in hemoglobin as well as the
level of rise in mean hemoglobin was significantly more in FS group at
follow up. The results are similar to that reported by Arvas, et al.(4)
and Langstaff, et al.(5). In contrast, both preparations were found
to cause equivalent increase in hemoglobin and serum iron levels by Sozmen,
et al.(6). In several other studies, the response to IPC was not
adequate(7-9). Though the gastrointestinal side effect are more in ferrous
sulfate group, yet the residual complaints were more in the iron
polymaltose groups.
Our study has thus demonstrated the superiority of FS
over IPC in treatment of IDA where hemoglobin rise as well as improvement
in constitutional symptoms was considered. Our conclusions need to be
substantiated in further randomized clinical trials on pediatric
population with a longer follow up.
Contributors: AVB was responsible for
acquisition of data, clinical examination and follow up interpretation of
data. RM edited the manuscript and statistically analyzed the data. GSP
conceived and designed the study, monitored the study and will act as
guarantor. RD reviewed the manuscript for important intellectual content
and approved the final draft.
Funding: None.
Competing interests: None stated.
|
What This Study Adds?
• Ferrous
sulfate has a better clinical response as compared to Iron
polymaltose complex for treating iron deficiency anemia in
children.
|
References
1. Nestel P, Alnwick D. Iron-micronutrient supplements
for young children. Summary and conclusions of a consultation held at
UNICEF, Copenhagen, August 19-20, 1996.
2. Borbolla JR, Cicero RE, Dibildox MM, Sotres DR,
Gutierrez RG. IPC vs. Iron sulphate in the treatment of iron
deficiency in infants. Rev Mex Padiatr 2000; 67: 63-67.
3. Jacobs P. Oral iron therapy in human subjects:
comparative absorption between ferrous salts and iron polymaltose. J Med
1984; 3: 387-377.
4. Arvas A, Gur E. Are ferric compound useful in
treatment of IDA? Turk J Pediatr 2000;42:352-353.
5. Langstaff RJ, Geisser P, Heil WG, Bowdler JM.
Treatment of IDA: a lower incidence of adverse effects with IPC than FS.
Brit J Clin Res 1993; 4: 191-198.
6. Sozmen EY, Kavakli K, Cetinkaya B, Akcay YD, Yilmaz
D, Aydinok Y. Effects of iron (II) salts and iron (III) complexes
on trace element status in children with iron-deficiency anemia. Biol
Trace Elem Res 2003; 94: 79-86.
7. Mehta BC. Ineffectiveness of iron polymaltose in
treatment of IDA. J Assoc Physicians India 2003; 51: 419-421.
8. Nielsen P, Gubbe EE, Fischer R, Heinrich HC.
Bioavailability of iron from ferric polymaltose in human. Drug Res 1994;
44: 743-748.
9. Heinrich HC. Intestinal iron absorption of 59 Fe
from neutron activated commercial iron (III) citrate and iron (III)
hydroxide polymaltose in humans. Drug Res 1987; 37: 105-107.
|
|
|
 |
|

