|
|
|
Indian Pediatr 2015;52:
245-246 |
 |
Kawasaki Disease with Autoimmune Hemolytic
Anemia
|
|
Dhwanee Thakkar, Nita Radhakrishnan,
*PK Pruthi and Anupam Sachdeva
From Pediatric Hematology Oncology and BMT Unit; and
*Institute of Child Health; Sir Ganga Ram Hospital, Rajinder Nagar, New
Delhi, India.
Correspondence to: Dr Anupam Sachdeva, Head of
Department, Institute of Child Health,
Sir Ganga Ram Hospital, Rajinder Nagar, New Delhi 110 060, India.
Email:
[email protected]
Received: July 30,2014;
Initial review: November 24, 2014;
Accepted: January 8, 2015.
|
|
Background: Association of
autoimmune haemolytic anaemia has been seldom reported with Kawasaki
disease. Case characteristics: A 7-month-old boy, presented with
prolonged fever, erythematous rash, severe pallor and hepatosplenomegaly.
Observations: Positive Direct Coombs test and coronary artery
aneurysm on echocardiography. He was managed with steroids along with
intravenous immunoglobulins and aspirin. Outcome: Early
identification of the condition helped in the management. Message:
Patients of autoimmune hemolytic anemia with unusual features such as
prolonged fever, skin rash, and mixed antibody response in Coombs test
should be evaluated for underlying Kawasaki disease as a possible
etiology.
Keywords: Autoimmunity, Coombs test, Coronary
artery aneurysm.
|
|
Autoimmune haemolytic anemia (AIHA) occurs in
children either secondary to infections, autoimmune conditions and
drugs, or may be primary [1]. Several hematological abnormalities have
been described in Kawasaki disease. AIHA is a rare association, and most
cases have occurred after infusion of intravenous immunoglobulins (IVIG)
in Kawasaki disease [2]. Simultaneous occurrence of AIHA with Kawasaki
disease has been very rarely observed [3-7].
Case Report
A 7-month-old boy presented with fever for 1 month
along with maculopapular rash all over the body with erythema of palms
and soles at the onset of illness. He was noted to have anemia and was
transfused thrice in the first one month of illness. Blood culture grew
Klebsiella pneumoniae and he was started on antibiotics
accordingly. As fever persisted and anemia progressed to cause cardiac
decompensation, he was referred to our center. Difficulty in
cross-matching of blood was noted at the blood bank of the referring
hospital. At the time of admission to our center, he had fever, severe
anemia, mild icterus, generalized lymphadenopathy and
hepato-splenomegaly. In addition, he had erythema of palms and soles,
and desquamation was noted on extremities. Hemoglobin was 3.6 g/dL,
white blood cell count 41.7×10 9/L
with 78% neutrophils, and platelet count was 407,000/µL; corrected
reticulocyte count was 3.4%. Peripheral smear examination showed
anisopoikilocytosis, polychromasia, neutrophilic leucocytosis with
monocytosis and few atypical lymphocytes. C-Reactive protein was 94
mg/L; Direct Coombs Test (DCT) was positive (3+) and showed a mixed
pattern: IgG 3+, IgM 3+, C3d 3+, IgA and C3c negative. Mycoplasma IgM
was positive. Polymerase chain reaction (PCR) for cytomegalovirus (CMV)
was 19362 copies/mL. PCR for Ebstein-Barr virus was negative. There was
no evidence of pneumonia on chest X-ray. Bone marrow examination
showed erythroid hyperplasia.
In view of the prolonged fever, erythematous rash and
desquamation of extremities, he was investigated for underlying Kawasaki
disease. 2D Echocardiography showed aneurysms of the main coronary
arteries; left main coronary artery 4.6 mm (Normal range 1.23-2.4 mm; Z
score 7.9); left anterior descending artery 3.97 mm (Normal range
1.06-1.76 mm; Z score 12.2); right coronary artery 3.87 mm (0.9-1.86 mm;
Z score 8.5) along with mild mitral regurgitation and good biventricular
function. A diagnosis of Kawasaki disease was made as per AHA guidelines
[8,9]. He received IVIG at 2 g/kg as a single dose, along with aspirin
(80 mg/kg/day). In view of AIHA, prednisolone was started at a dose of 2
mg/kg/day along with supportive transfusion therapy. Azithromycin was
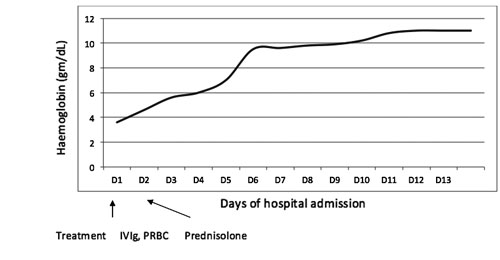
given for 5 days. Child became afebrile within 24 hours and hemoglobin
remained stable after transfusion (Fig. 1). He was
discharged after 4 days of admission on aspirin and steroids. CMV PCR
was repeated which was negative, and hence no definitive treatment for
the same was given. Dose of steroids is at present being tapered slowly
and aspirin is being continued at antiplatelet dose. Clinically there is
regression of lymphadenopathy and hepatosplenomegaly. He is being kept
under close follow up for monitoring the size of coronary aneurysms.
 |
|
Fig. 1 Trend of hemoglobin rise in a
child with Kawasaki disease and autoimmune hemolytic anemia.
|
Discussion
Hematological abnormalities known to occur with
Kawasaki disease include neutrophilic leucocytosis, thrombocytosis and
normocytic anemia. The association of AIHA with Kawasaki disease has
been rarely observed. In the index case reported by Kawasaki, the
patient had fall in hemoglobin and positive DCT at multiple occasions.
However, since no other patient in the original series had similar
findings, this was not reported in his landmark English publication [3].
Reticulocyte response was not as brisk as expected
for the degree of hemolysis in our case. AIHA has been reported to have
reticulocytopenia due to the autoimmune destruction of RBC precursors
and increased apoptosis [1]. The pattern of antibodies as observed by
monospecific DCT in our case was also unusual. Usually the antobody
pattern observed in AIHA is IgG in warm antibody-mediated AIHA and
paroxysmal cold hemoglobinuria, and IgM in cold agglutinin disease, with
respective thermal amplitudes. A mixed pattern was observed in our case
with 3+ positivity for IgG, IgM and C3d. Mixed type AIHA account for a
minor percentage of the total AIHA cases in children, and is seen
usually secondary to drugs and rheumatological conditions [1,4].
In most cases of AIHA with Kawasaki disease, it has
been reported as a complication following IVIG infusion [2]. IVIG can
produce hemolysis directly by isohemagglutinins or by stimulating B
lymphocytes resulting in the production of RBC autoantibodies [6]. In
our patient, onset of AIHA occurred prior to IVIG administration ,
ruling out this possibility. The reasons for AIHA in our patient
includes any one or a combination of the underlying rheumatological
condition (Kawasaki disease), and infections such as Mycoplasma, CMV or
Klebsiella – all of which were detected at various time points.
The resolution of hemolysis in our patient coincided with the resolution
of Kawasaki disease. Clinical response and improvement was seen after
starting steroids along with IVIG and aspirin.
We conclude that patients of AIHA with unusual
features such as prolonged fever, skin rash, or mixed antibody response
in DCT should be evaluated for underlying rheumatologic diseases such as
Kawasaki disease. Early identification and management will help in
reducing complications of both problems.
Contributors: All authors were involved in all
aspects of management and manuscript preparation.
Funding: None. Competing interests: None
stated.
References
1. Ware RE. Autoimmune Hemolytic Anemia. In:
Orkin SH, Nathan DG, editors. Hematology of Infancy and Childhood.
7th ed. Philadelphia: Saunders Elsevier; 2009. p. 614-634
2. Huang FL, Chang TK, Jan SL, Tsai CR, Wang LC,
Lai MC, et al. Co-morbidity of Kawasaki disease. Indian J
Pediatr. 2012;79:815-7.
3. Shulman ST. Hemolysis in Kawasaki disease.
Transfusion. 1991;31:572.
4. Panzarino V, Estrada J, Benson K, Postoway N, Garratty
G. Autoimmune hemolytic anemia after Kawasaki disease in a child.
Int J Hematol. 1993; 57:259-63.
5. Bunin NJ, Carey JL, Sullivan BD. Autoimmune
hemolytic anemia in association with Kawasaki disease. Am J Pediatr
Hematol Oncol. 1986;8:351-3.
6. Hillyer CD, Schwenn MR, Fulton DR, Meissner
HC, Berkman EM. Autoimmune hemolytic anemia in Kawasaki disease: a
case report. Transfusion. 1990;30:738-40.
7. Cataldo F, Violante M, Bellia L, Gueci G,
Maltese I, Albeggiani A. Autoimmune hemolytic anemia in the course
of Kawasaki disease. A case presentation. Minerva Pediatr.
1991;43:6614.
8. Newburger JW, Takahashi M, Gerber MA, Gewitz
MH, Tani LY, Burns JC, et al. Diagnosis, treatment, and
long-term management of Kawasaki disease: A statement for health
professionals from the committee on Rheumatic Fever, Endocarditis,
and Kawasaki Disease, Council on Cardiovascular Disease in the
Young: American Heart Association. Pediatrics. 2004;114:1708-33.
9. Dallaire F, Foumier A, Breton J, Nguyen TD, Spigelblatt L, Dahdah
N. Marked variations in serial coronary artery diameter measures in
Kawasaki disease: A new indicator of coronary involvement. J Am Soc
Echocardiogr. 2012;25:859-65.
|
|
|
 |
|

