Hemorrhagic pleural effusions in
childhood are commonly associated with tuberculosis, trauma,
malignancies and collagen vascular disease(1). We present here a case of
massive "hemorrhagic pleural effusion of a rare
etiology.
Case Report
A 9-year-old male child presented with a
12 day history of fever and dry cough. Two days prior to admission, the
patient developed pleuritic pain over the right hemithorax. A history of being kicked in
the abdomen by a school-mate about 2 months ago followed by mild
abdominal pain was obtained later during the course of his hospital
stay.
Examination revealed an undernourished
febrile child with features of rightsided pleural effusion. Examination
of the abdomen was normal. Chest X-ray confirmed the clinical finding of
a right-sided pleural effusion. The pleural fluid was uniformly
hemorrhagic and contained 8.8x109 leukocytes, predominantly
neutrophils, 4.8 g/dl proteins, 87 mg/dl glucose, no malignant cells and Was sterile on culture. The ESR was 48
mm/1st hour and the Mantoux test was negative.
He was initially started on intravenous
antibiotics (cloxacillin and gentamicin). He became afebrile within 4
days and oral cloxacillin was continued for 4 weeks. However, his
effusion persisted and reaccumulated rapidly after a therapeutic
drainage (350 ml). An underlying tubercular etiology. was considered and
he was put on anti-tubercular drugs (ATT).
Inspite of A IT the effusion persisted
and required an intercostal tube drain. Initially, the drainage was
around 100 ml/day, but gradually decreased to 10-20 ml/day over 10
days. Chest X-ray done at this time showed good lung expansion. An
ultrasound of the chest done to look for any residual fluid
showed, in addition to the right pleural
effusion, an anechoic collection in the region of the pancreas measuring
4.5x2.1 cm suggestive of a pancreatic psuedocyst with a communication to
the pleural space (pancreaticopleural fistula) (Fig.1). Serum
amylase was 47 Somogyi units and pleural fluid amylase 400 Somogyi
units. CT scan abdomen showed 2 intrapan-creatic pseudocysts (Fig.
2), one of which was extending into the mediastinum and right
pleural space (Fig. 3). Bilateral pleural effusions (R>L) were
also seen.
He was managed conservatively with right
intercostal tube drainage for 14 days. ATT
was stopped. During his hospital stay, he developed super added
infection of pleural fluid with pseudomonas aeruginosa which
responded to parenteral antibiotics administered for 7 days. The child
improved symptomatically and the pleural effusion resolved. He remained
asymptomatic and was discharged after about 2 months of hospital stay.
On follow up after 21/2 months, he remained asymptomatic and
one ultrasound abdomen showed reduction in the cyst size. Subsequently
he was lost to follow up.
 |
|
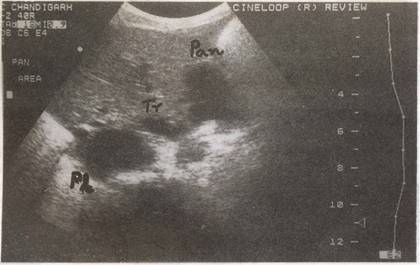
Fig. 1. Ultrasound abdomen showing pancreatic cyst
(Pan) and pleural collection (PI) with the communicating
tract (Tr). |
 |
|
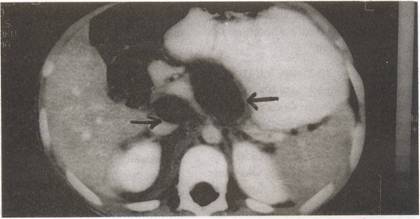
Fig. 2. CT scan
abdomen showing two pancreatic
pseudocysts. |
 |
|
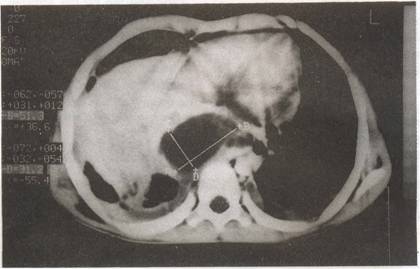
Fig. 3. CT scan chest showing mediastinal pseudocyst and
bilateral pleural effusions (R>L). |
Discussion
Tuberculous pleural effusion is a common cause of hemorrhagic pleural
effusion in India. In the absence
of an obvious cause, patients with hemorrhagic pleural effusion have
been empirically started on ATT as was done in our patient(2).
Pancreatic pseudocysts have been
infrequently reported in the pediatric population(3).
Pancreaticopleural fistula is an uncommon complication that occurs in
1-3% of cases of pancreatic pseudocysts in adults(4,5). However, the
incidence in children is not known. Blunt abdominal trauma accounts for
over 60% of the reported cases of pancareatic pseudocysts in
children(3).
Pancreatic pleural effusions result from
either a posterior disruption of the duct or a leaking pseudocyst(4-6).
Spillage of pancreatic secretions into the retroperitoneum occurs and these track along the aortic or esophageal hiatus into the
mediastinum. The secretions may either get localized to form a
mediastinal pseudocyst or rupture into one or both pleural cavities as
pleural effusions. Anterior pancreatic ductal disruption, on the other
hand, leads to pancreatic ascites(4,5).
While a diagnosis of pleural effusion of
a pancreatic origin is based on a high amylase concentration(2,4-6)
demonstration of pancreatic pseudocysts and pancreaticopleural fistula
requires radiologic investigations(4). Ultrasound is useful in defining
pancreatic pseudocysts; however CT scan is better in defining pancreatic
abnormality and can often demonstrate pancreatic pseudocysts with direct
extension into the pleural cavity(4,5). ERCP plays an important role in
defining the ductal anatomy and fistula and is essential before surgical
intervention( 4).
