|
|
|
Indian Pediatr 2017;54: 112-115 |
 |
Effect of Delayed Cord
Clamping on Hematocrit, and Thermal and Hemodynamic Stability in
Preterm Neonates: A Randomized Controlled Trial
|
|
Niraj Kumar Dipak, Ruchi Nimish Nanavati, Nand
Kishore Kabra, Anita Srinivasan and Anitha Ananthan
From Department of Neonatology, Seth GS Medical
College and KEM Hospital, Parel, Mumbai, India.
Correspondence to: Dr Niraj Kumar Dipak, Department
of Neonatology, Seth GS Medical College and KEM Hospital, Acharya Donde
Marg, Parel, Mumbai 400 012, Maharashtra, India.
Email: [email protected]
Received: January 23, 2016;
Initial review: March 26, 2016;
Accepted: December 22, 2016.
|
Objective: To evaluate the short term clinical
effects of delayed cord clamping in preterm neonates.
Design: Randomized controlled trial.
Setting: A tertiary care neonatal unit from
October 2013 to September 2014.
Participants: 78 mothers with preterm labor
between 27 to 316/7 weeks gestation.
Intervention: Early cord clamping (10 s), delayed
cord clamping (60 s) or delayed cord clamping (60 s) along with
intramuscular ergometrine (500 µg) administered to the mother.
Main outcome measures: Primary: hematocrit
at 4 h after birth; Secondary: temperature on admission in
neonatal intensive care unit, blood pressure (non-invasive) at 12 h, and
urinary output for initial 72 h.
Results: Mean (SD) hematocrit at 4 h of birth was
58.9 (2.4)% in delayed cord clamping group, and 58.7 (2.1)% in delayed
cord clamping with ergometrine group as compared to 47.6 (1.3)% in early
cord clamping group. Mean (SD) temperature on admission in NICU was 35.8
(0.2)ºC, 35.8 (0.3)ºC, and 35.5 (0.3)ºC, respectively in these three
groups. The mean (SD) non-invasive blood pressure at 12 h of birth was
45.8 (7.0) mmHg, 45.8 (9.0) mmHg, and 35.5 (8.6) mmHg, respectively in
these three groups. Mean (SD) urinary output on day 1 of life was 1.1
(0.2) mL/kg/h, 1.1 (0.2) mL/kg/hr and 0.9 (0.2) ml/kg/h, respectively.
Conclusion: In preterm neonates delayed cord
clamping along with lowering the infant below perineum or incision site
and administration of ergometrine to mother has significant benefits in
terms of increase in hematocrit, higher temperature on admission, and
higher blood pressure and urinary output during perinatal transition.
Keywords: Anemia, Hypothermia, Newborn resuscitation,
Umbilical cord.
|
|
O
ptimal cord clamping time in premature neonates
remains controversial [1].
Delayed cord clamping (DCC) is considered a more physiological mechanism
by which the newborn may receive an additional blood volume to perfuse
the lungs, intestines, kidneys and the skin, favoring successful
adaptation to the extrauterine life [2], and has been recently
recommended as preferred method of cord clamping in preterm neonates,
when feasible [3]. However, other birth-related factors also influence
the speed and amount of placental transfusion at birth; the level at
which the infant is held; the type and method of delivery; uterine
contractions during third stage; and administration of uterotonics (oxytocin
or ergometrine) [4]. Yao, et al.
[5] demonstrated that a strong uterotonic drug
administered intravenously to the mother increased the rate of placental
transfusion without a risk of over transfusion. We conducted this single
center randomized controlled trial to test the primary hypothesis that
for preterm neonates, lowering the infant below the perineum/incision
site, administration of intramuscular ergometrine to the mother and
delayed cord clamping will result in higher hematocrit and improved
thermal and hemodynamic stability.
Methods
The study was conducted over a period of 12 months
(October 2012 to September 2013) at a tertiary care hospital in Mumbai,
India. The study was approved by institutional ethics committee of the
hospital. Mothers with 27-31 6/7
weeks’ gestation with preterm onset of labor were
included to participate in the study. Mothers with multiple gestation,
Rh-ve status, placenta previa or abruption-placenta, and those having
fetus with major congenital anomalies, hydrops, fetal growth restriction
with abnormal Doppler waveforms, or evidence of foetal distress were
excluded from the study. Mothers, who fulfilled the inclusion criteria,
when they came in labor, were enrolled in the study during onset of
labor after obtaining informed consent. Mothers were assigned to Early
cord clamping (ECC) group, Delayed cord clamping group 1(DCC1) or
Delayed cord clamping group 2E (DCC 2E). For the ECC group, obstetrician
clamped the umbilical cord at 10 seconds and baby was held supine at
level of introitus/placental incision. For the DCC1 group, neonates were
held in a pre-warmed towel approximately 10-15 inches below the
introitus at vaginal delivery/below the level of placental incision in
caesarean delivery, and cord was clamped at 60 seconds. For the DCC2E
group, neonates were held 10-15 inches below the introitus at vaginal
delivery/below the level of placental incision in caesarean delivery,
injection ergometrine 500 µg intramuscular (IM) was administered to the
mother, and cord was clamped at 60 seconds. A stopwatch was used to mark
the time that was counted in 10 seconds interval. Allocation of groups
was done by random number sequence with variable block size of 3 or 6
using a ‘Random Allocation Software’ program. The sequence was concealed
in serially numbered, opaque, sealed and identical envelopes. The random
allocation sequence was generated by a statistician who was not a part
of the study.
Antenatal and delivery details were entered in
mother’s chart. Umbilical cord blood was collected for blood gas
analysis within 30 minutes of collection. Timing of cord clamping, APGAR
score at 1 min and 5 min, and time of birth were recorded. Axillary
temperature was recorded with a mercury thermometer in labor room at
approximately 5 minutes.
After stabilization in labor room, neonates were
shifted to neonatal intensive care unit (NICU) and managed as per
standard protocol. At 4 hours of age, venous sample was collected for
hematocrit measurement. At 12 hour, heart rate, mean non-invasive BP
(NIBP), Clinical Risk Index for Babies (CRIB) score, max FiO 2
requirement, respiratory support during initial 24 hours, requirement of
surfactant, and arterial/alveolar ratio at 24 hour were recorded in all
neonates. For initial 72 hours, urinary output was recorded. At 7, 14
days and 40 weeks post-menstrual age, neurosonogram was performed.
Neonates were followed-up for retinopathy of prematurity (ROP) screening
and subsequent retinal examinations.
Primary outcome was hematocrit at 4 hours of age.
Secondary outcomes were temperature on admission, heart rate, NIBP at 12
hours, urinary output for initial 72 hours, number of red cell
transfusions, total serum bilirubin (TSB) at 72 hours, peak serum
bilirubin (PSB), evidence of retinopathy of prematurity (ROP),
intra-ventricular hemorrhage (IVH), late onset sepsis (LOS), and
nectrozing enterocolitis (NEC) stage 2 or more.
Sample size calculation was based on venous
hematocrit at 4 hours of age in immediately clamped infants. Considering
it as [48(4) % SD] [5,6] and using initial venous hematocrit as the
primary outcome variable and an expected 10 to 15% relative increase by
DCC with an alpha error 0.05 and power 80%, we estimated the need to
enrol 30 neonates in each group. Statistical analysis was performed
using SPSS version 16. Data were analyzed by intention to treat
principle. P<0.05 was considered as statistically significant.
Results
Flow of recruitment of study participants is shown in
(Fig. 1).The demographic profile was comparable in all the
three groups (Table I).
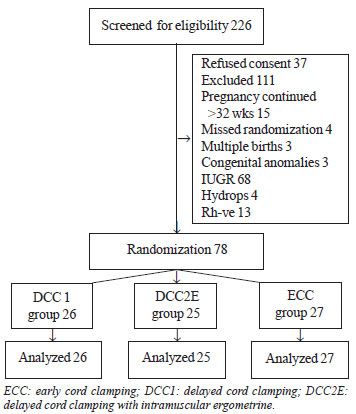 |
|
Fig. 1 Flow of recruitment of study
participants.
|
TABLE I Comparison of Baseline Characteristics in the Three Study Groups
|
Maternal and infant characteristics |
ECC(n=27) |
DCC1(n=26) |
DCC2E(n=25) |
|
Mother’s age (y) |
26.6 (4.2) |
26.6 (3.9) |
26.0 (4.2) |
|
*Gestational age (wk) |
29.9 (1.4) |
30.1(1.2) |
30.2 (1.2) |
|
Caesarian delivery |
4 (14.8) |
4 (15.4) |
4 (16) |
|
Chorioamnionitis |
3 (11.1) |
3 (11.5) |
2 (8) |
|
Birth Weight (g) |
1283.7 (176.4) |
1316.1 (162.9) |
1297.9 (177.8) |
|
*Baseline Temperature at 5 min after birth (ºC) |
34.0 (0.7) |
33.9 (0.8) |
34.2 (0.5) |
|
*Resuscitation required |
7 (26%) |
8 (31%) |
6 (24%) |
|
*Cord blood pH |
7.2 (0.1) |
7.2 (0.1) |
7.2 (0.1) |
|
ECC: early cord clamping; DCC1: delayed cord clamping; DCC2E:
delayed cord clamping with intramuscular ergometrine. Values in
n (%) or *mean (SD). |
Mean (SD) venous hematocrit at 4 hours was 47.6
(1.3)% in ECC group as compared to 58.9 (2.5)% in DCC1 and 58.7 (2.1)%
in DCC2E group. There was statistically significant mean (SD) difference
in venous hematocrit in DCC1 group when compared with ECC group, and
when DCC2E was compared with ECC group (Table II). In ECC
group, 6 infants required vasopressor support, while in DCC1 group and
DCC2E, 1 and 2 infants, respectively required vasopressor support.
TABLE II Comparison of Outcome Measures Between the Three Groups
|
Characteristics |
Post Hoc multiple comparison ,weighted |
Post hoc multiple comparison, |
|
mean difference (SD) between groups ECC |
weighted mean difference (SD) |
|
and DCC1(P value) |
between groups ECC and DCC2E |
|
|
( P value) |
|
Venous hematocrit at 4 hours |
11.3 (0.6) (<0.001) |
11.1 (0.6) (<0.001) |
|
Admission temperature |
0.3 (0.1) (0.004) |
0.3 (0.1) (0.008) |
|
Mean BP at 12 hours (mm hg) |
10.2 (2.3) (<0.001) |
10.3 (2.3) (<0.001) |
|
Urine Output in first 24 hours (ml/kg/hr) |
0.3 (0.1) (<0.001) |
0.2(0.1) (<0.001) |
|
Urine Output in next 24 hours (ml/kg/hr) |
0.7 (0.1) (<0.001) |
0.7 (0.1) (<0.001) |
|
TSB at 72 hours (mg/dL) |
3.8 (0.8) (<0.001) |
3.8 (0.8) (<0.001) |
|
PSB (mg/dL) |
2.1 (0.9) (0.02) |
2.5 (0.9) (0.01) |
|
*TSB: Total serum bilirubin , # PSB : Peak serum bilirubin;
ECC: early cord clamping; DCC1: delayed cord clamping; DCC2E:
delayed cord clamping with intramuscular ergometrine. |
Among three groups, 11 (40.7%), 2 (7.7%), 1 (4%) in
ECC, DCC1 and DCC2E groups, respectively required red cells transfusion.
Mean (SD) TSB at 72 hours was 9.4 (3.1) mg/dL in DCC1 and 9.4 (3.3) mg/dL
in DCC2E as compared to 5.6 (1.7) mg/dL in ECC group. In ECC group,
10/27 (37%) had evidence of brain injury while 6/26 (23.1%) in DCC1
group and 5/25 (20%) in DCC2E had evidence of brain injury. None of the
neonates in DCC group required therapy for ROP while 2 (7.4%) babies in
ECC group underwent laser phototherapy. Nine (33.3%) babies in ECC
group, 5 (19.2%) in DCC1 and 3 (12%) babies in DCC2E group developed
late onset sepsis. Evidence of hemodynamically significant patent ductus
arteriosus (hsPDA) was seen in 5 (18.5%), 4 (15.4%) and 2 (8%) neonates
in ECC, DCC1 and DCC2E groups, respectively.
There was no significant difference among three
groups for maximum FiO 2
requirement in first 12 hours, heart rate at 12 hours, respiratory
support needed, surfactant administration, CRIB score in first 12 hours,
a/A ratio, arterial blood pH, base deficit at 12 hours, LOS during NICU
stay, evidence of brain injury on USG skull, ROP needing treatment or
presence of hsPDA requiring medical closure.
Discussion
In this randomized controlled trial, higher venous
hematocrit values at 4 h of age in delayed clamping groups indicate that
delayed cord clamping is effective in increasing the placental
transfusion in preterm neonates. Infants in delayed clamping group had
improved temperature on admission in NICU in our study but there was no
difference between DCC1 and DCC2E group.
One of the limitations of this study was that we did
not measure effects of delayed cord clamping on blood volume. However,
we checked the indirect manifestations of increased blood volume such as
initial hematocrit, blood pressure and urinary output. Recording of only
short-term effects of delayed clamping could be another limitation.
Outcomes of delayed cord clamping were not studied in cases of growth
retarded babies and in non-vigorous neonates requiring resuscitation.
Apart from timing of cord clamping, position of infants and use of
ergometrine after delivery, other factors which affect the placental
transfusion i.e. mode of delivery (vaginal versus caesarean),
were not compared. Single-center based study and small sample size were
other limitations.
Finding of raised hematocrit is consistent with
observations of Ibrahim, et al. [8], who reported that even 20
seconds’ delay in cord clamping could cause rise in hematocrit at 4
hours. Oh, et al. [9] also documented that a delay of 30-45
seconds caused rise of hematocrit. However, Mc Donnel, et al.
[10] found no difference in hematocrit at 4 hours in relation to timing
of cord clamping. A Cochrane systemic review [7] concluded that DCC is
associated with fewer transfusion requirements for anemia; for every 100
babies subjected to delayed cord clamping, 27 are spared a blood
transfusion. Further studies with large sample size are required to
evaluate whether delayed cord clamping in preterm neonates leads to low
incidence of complications such as late onset sepsis, necrotizing
enterocolitis, bronchopulmonary dysplasia or retinopathy of prematurity.
Studies can be undertaken for considering the possibilities of delayed
clamping in preterm neonates who require immediate resuscitation so that
both resuscitation and delayed cord clamping can occur simultaneously.
We conclude that in preterm infants, the placental transfusion achieved
with lowering the infant below perineum or incision site, administration
of intramuscular ergometrine and delayed cord clamping by 60 seconds, is
associated with less hypothermia on admission in NICU and improved blood
pressure and increased urinary output during the perinatal transition.
Contributors: NKD, AS, AA: were involved in
management of patient, designing the study and drafting the manuscript;
RNN, NKK: were involved in the review of literature, and manuscript
preparation. The final manuscript was approved by all the authors.
Funding: None; Competing interest: None
stated.
|
What is Already Known?
• Delayed cord clamping in preterm neonates
is associated with improved hematocrit and less incidence of
anemia at 6-10 weeks of age.
What This Study Adds?
• This study demonstrates the cumulative
effects of (i) lowering the infant position by 10-15 cm
below the perineum/incision site; (ii) administration of
inj ergometrine; in addition to (iii) delayed cord
clamping on placental transfusion.
• This better placental transfusion, is
associated with less hypothermia on admission in NICU and
improved blood pressure and urinary output during the perinatal
transition.
|
References
1. Van Rheenen P, De Moor L, Eschbach S, De Grooth H,
Brabin B. Delayed cord clamping and haemoglobin levels in infancy: A
randomised controlled trial in term babies. Trop Med Int Health.
2007;12:603-16.
2. Farrar D, Tuffnell D, Airey R, Duley L. Care
during the third stage of labour: A postal survey of UK midwives and
obstetricians. BMC Pregnancy Childbirth. 2010;10:23.
3. Tarnow-Mordi WO, Duley L, Field D, Marlow
N, Morris J, Newnham J, et al. Timing of cord clamping in
very preterm infants: More evidence is needed. Am J Obstet Gynecol.
2014;211:118-23.
4. Yao AC, Moinian M, Lind J. Distribution of
blood between the infant and the placenta after birth. Lancet.
1969;7626:871-3.
5. Lindercamp O, Nelle KM, Zilow EP. The effect of
early and late cord clamping on blood viscosity and other
hemorheological parameters in full term neonates. Acta Paediatrica.
1992;81:45-750.
6. Obladen M, Diepold K, Maier RF. The European
Multicentre rhEPO study group: Venous and arterial haematological
profiles of very low birth weight infants. Pediatrics. 2000;106:707-9.
7. Rabe H, Diaz-Rossello JL, Duley L, Dowswell T.
Effect of timing of umbilical cord clamping and other strategies to
influence placental transfusion at preterm birth on maternal and infant
outcomes. Cochrane Database Syst Rev. 2012:8:CD003248.
8. Ibrahim HM, Krouskop RW, Lewis DF, Dhanireddy R.
Placental transfusion: Umbilical cord clamping and preterm infants. J
Perinatol. 2000;20:351-4.
9. Oh W, Fanaroff AA, Carlo WA, Donovan EF, Mc Donald
SA, Poole WK. Effects of delayed cord clamping in very low birth weight
infants. J Perinatol. 2011;31:568-71.
10. McDonnell M, Henderson-Smart DJ. Delayed umbilical cord clamping
in preterm infants: a feasibility study. J Paediatr Child Health.
1997;33:308-10.
|
|
|
 |
|

