|
|
|
Indian Pediatr 2014;51: 128-130 |
 |
Heparin Co-factor II Thrombin Complex as a
Biomarker for Mucopolysaccharidosis:
Indian Experience
|
|
Sanjeev Kumar Pandey, Ankur Singh, AP Dubey, *TK Mishra and Seema Kapoor
From Departments of Pediatrics and *Biochemistry, MAMC
and Associated Lok Nayak hospital, New Delhi, India.
Correspondence to: Dr Seema Kapoor, M-439,
Ground Floor, Guruharkishan Nagar,
Paschim Vihar, New Delhi, India.
Email: drseemakapoor@gmail.com
Received: April 01, 2013;
Initial review: May 15, 2013;
Accepted: August 22, 2013.
Published online: September 05, 2013.
PII: S097475591300319
|
Background: Serum heparin cofactor II-thrombin complex (HCII-T)
is an emerging biomarker for mucopolysaccharidosis disease (MPS I
and MPS II). Methods: Seventeen cases (6 MPS I and 11 MPS II)
and sixty healthy controls were enrolled in study, conducted from
September 2008 to December 2012. The mean ± SD age of MPS1 (n=6, 5
males) and MPS II was 7.02 ± 3.25 and 5.2 ± 2.15 years,
respectively. Disease status was confirmed by clinical features and
enzyme assay. Urinary glycosaminoglycans were measured in spot urine
samples and expressed in relation to creatinine content. HCIIT
measurement was done using sandwich ELISA at enrolment and after 12
and 24 months of recruitment. Results: Urinary
glycosaminoglycans and HCIIT were elevated in all patients compared
to their healthy controls. Both markers could not discriminate
between the type of mucopolysaccharidosis. Conclusion:
Heparin Cofactor II Thrombin Complex is a good biomarker for
mucopolysaccharidosis I and II.
Keywords: Diagnosis, Glucosamino-glycans,
Mucopolysaccharidosis, Screening
|
|
The mucopolysaccharidosis (MPS)
are progressive, multisystemic disorders that are caused by genetic
defects in the catabolism of glycosaminoglycans (GAG) [1,2]. Considering
the complexity of the disease and the advent of enzyme replacement
therapy for both these disorders [3-5], there exists a need for a robust
biomarker. Serum Heparin Cofactor II Thrombin has emerged as a biomarker
for certain subtypes of MPS. Considering the scarcity of literature and
lack of Indian studies, we studied its role in MPS l and MPS II [6].
Methods
After approval from Institute Ethical Committee, the
study was conducted from September 2008 to December 2012. Seventeen
cases (6 MPS I and 11 MPS II) and 60 healthy controls were enrolled in
the study after getting informed consent. Disease status was determined
based on clinical presentation and enzyme assay on dried blood spot
[7,8]. Blood samples were collected with sterile technique from both
cases and controls and serum was separated and stored at –80 degree
Celsius till analysis. Urinary glycosaminoglycans were estimated in
early morning samples, as 24-hour collection is difficult in pediatric
age group and more so in neurologically impaired children. Prior to
this, normative age stratified data had been established in the
laboratory for our population. The estimation was done by using
Dimethylmethylene blue as the binding reporter dye [9]. HCII T was
measured using ELISA method from commercially available kits (Affinity
Biologicals, Hamilton, ONUS). Both urine and blood samples were
collected thrice; at start of enrolment, one year after enrolment, and
two years after enrolment.
Results
The mean (SD) age of MPS I (n=6, 5 males) and
MPS II (n=11, all males) patients was 7.02 (3.25) and 5.2 (2.15)
years, respectively. Spot urinary GAG were measured and were stratified
age-wise. This was the first investigation used in a case suspected with
MPS. Urinary GAG were markedly elevated in all patients with no
significant change in between MPS I and MPS II. The median (interquartile
range) HCII T in MPS I and MPS II was 455 (350-500) ng/mL and 340 (265-
530) ng/mL, respectively, as compared to 39 (12-60) ng/mL in normal
children. No significant difference was observed in levels of HCII-T
over the next 2 years (Fig.1). One patient who had access
to therapy showed decline in values with follow up: at start of therapy
(330 ng/mL), 18 months (215 ng/mL), and at 28 months (135 ng/mL) after
therapy.
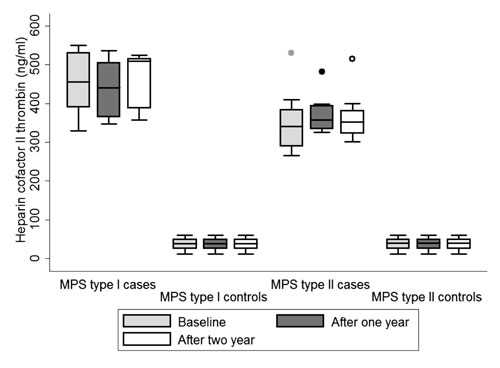 |
|
Fig.1 Box plot showing Heparin
Cofactor II Thrombin levels in controls and MPS Type I and MPS
Type II patients at 1 st visit
(Baseline) and at different time intervals.
|
Discussion
(HCII T) and urinary glycosaminoglycans were elevated
in both MPS I and MPS ll patients as compared to controls at the
beginning of enrolment. Follow up levels of these two biomarkers at end
of one year and two year showed consistently high levels from previous
ones, highlighting the increase in disease burden status. Both these
biomarkers were unable to predict the type of MPS.
Glycosaminoglycans are widely used primary biomarkers
in MPS but have certain limitations. Estimation of GAG depends widely on
age, hydration and kidney status of affected cases. There is also a wide
range of normalcy in infancy. Another drawback is the need for 24-hour
urinary collection, which is difficult in uncooperative children.
Advantages of HCII-T include: one time collection of blood, estimation
independent of renal function, and less time consuming. Likely
disadvantage with HCII-T include: invasive procedure, expensive and
inability to differentiate among MPS I, MPS II and MPS VI as it is
elevated in all conditions with accumulation of dermatan sulphate.
We found a significant difference in the levels of
HCII-T found in healthy controls with compared to the disease group, as
reported previously [6, 11-13].
Regarding ability to adequately predict the course of
disease, we found increase in level of both biomarkers in both group of
patients without any significance. The plausible reason for this could
have been the short duration of follow up or inclusion of attenuated
phenotype, which may demonstrate slow progression and wide variability
in the course of disease. We found a fall in both urinary biomarker (glycosaminoglycans)
and blood biomarker (HCII-T) values in one patient at end of 18 and 28
months of initation of therapy, it is difficult to derive any conclusion
regarding comparative efficacy of the biomarkers from this single case.
To conclude, HCII-T is a reliable screening biomarker
for MPS. However, considering the limitation to differentiate between
subtypes and to predict the course in attenuated phenotypes, we need to
move from organ- or pathway-specific biomarkers to a panel of biomarkers
to correlate with prediction, typification, disease progression and
response to therapy.
Contributors: SKP: was involved in planning the
study and drafting the manuscript; AS: was involved in case enrolment
and follow up of cases; APD: guidance at various satges of study; TKM:
was involved in helping and guiding the laboratory work; and SK:
critically reviewed the manuscript, made the diagnoses and will act as
guarantor for the manuscript.
Funding: DBT; Competing interests: None
stated.
|
What This Study Adds?
• Heparin Cofactor II Thrombin Complex is a
good biomarker for Mucopolysaccharidosis I and II.
|
References
1. Whitley CB. The mucopolysaccharidoses. In:
Beighton P -McKusick’s Heritable Disorders of Connective Tissue. 5th
ed., St Louis, Mosby, 1993. P. 367-499.
2. Neufeld EF, Muenzer J. The mucopolysaccharidoses.
In: Scriver CR, Beaudet AL, Sly WS, Valle D, Childs B, Kinzler
KW, et al, editors. The Metabolic and Molecular Bases of
Inherited Disease. Vol. III. 8th ed. New York: McGraw-Hill; 2001.
P . 3421-3452.
3. Wraith JE, Clarke LA, Beck M, Kolodny EH, Pastores
GM, Muenzer J, et al. Enzyme replacement therapy for
mucopolysaccharidosis I: a randomized, double-blinded,
placebo-controlled, multinational study of recombinant human alpha-L-iduronidase
(laronidase). J Pediatr. 2004; 144:581-8.
4. Wraith JE. Enzyme replacement therapy with
idursulfase in patients with mucopolysaccharidosis type II. Acta
Paediatr Suppl. 2008; 97:76-8.
5. Harmatz P, Giugliani R, Schwartz IV, Guffon N,
Teles EL, Miranda MC, et al. MPS VI Study Group.Long-term
follow-up of endurance and safety outcomes during enzyme replacement
therapy for mucopolysaccharidosis VI: Final results of three clinical
studies of recombinant human N-acetylgalactosamine 4-sulfatase. Mol
Genet Metab. 2008;94:469-75.
6. Randall DR, Colobong KE, Hemmelgarn H, Sinclair
GB, Hetty E, Thomas A, et al. Heparin cofactor II-thrombin
complex: a biomarker of MPS disease.Mol Genet Metab. 2008; 94:456-61.
7. Tolun AA, Graham C, Shi Q, Sista RS, Wang T,
Eckhardt AE, et al. A novel fluorometric enzyme analysis method
for Hunter syndrome using dried blood spots.Mol Genet Metab.
2012;105:519-21.
8. Gusina NB, Tsukerman GL. Fluorometric
determination of alpha-L-iduronidase activity in leukocytes and blood
plasma in Hurler’s disease. Lab Delo. 1988;7:46-9.
9. Whitley CB, Draper KA, Dutton CM, Brown PA,
Severson SL, France LA. Diagnostic test for mucopolysaccharidosis. II.
Rapid quantification of glycosaminoglycan in urine samples collected on
a paper matrix. Clin Chem. 1989; 35:2074-81.
10. Schwartz IV, Ribeiro MG, Mota JG, Toralles MB,
Correia P, Horovitz D, et al. A clinical study of 77 patients
with mucopolysaccharidosis type II. Acta Paediatr Suppl. 2007; 96:63-70.
11. Randall DR, Sinclair GB, Colobong KE, Hetty E,
Clarke LA. Heparin cofactor II-thrombin complex in MPS I: a biomarker of
MPS disease. Mol Genet Metab. 2006; 88:235-43.
12. Langford-Smith K, Arasaradnam M, Wraith JE, Wynn
R, Bigger BW. Evaluation of heparin cofactor II-thrombin complex as a
biomarker on blood spots from mucopolysaccharidosis I, IIIA and IIIB
mice. Mol Genet Metab. 2010; 99:269-74.
13. Clarke LA, Winchester B, Giugliani R,
Tylki-Szymañska A, Amartino H. Biomarkers for the mucopolysaccharidoses:
discovery and clinical utility. Mol Genet Metab. 2012; 106:395-402.
|
|
|
 |
|

