|
|
|
Indian Pediatr 2014;51: 1003-1005 |
 |
Hematological Parameters in Adolescents with
Hyperuricemia
|
|
Marian J Stelmach, Lukasz Szczerbinski, Natalia
Wasilewska, Piotr Protas and Anna Wasilewska
From Department of Pediatric Nephrology, Medical
University of Bialystok, Poland.
Correspondence to: Dr Marian Stelmach, Department of
Health and Social Sciences, Pope John Paul II State School of Higher
Education, 95/97 Sidorska Street, 21-500 Biala Podlaska, Poland.
Email: [email protected]
Received: March 14, 2014;
Initial review: May 08, 2014;
Accepted: October 01, 2014
|
Objective: To examine the association between hematological
indices and serum uric acid in adolescents with hyperuricemia.
Methods: 10-year retrospective cohort study of 607 patients with
hyperuricemia registered with the Pediatric Nephrology Department.
Results: There was a statistically significant positive
correlation between serum uric acid levels and BMI Z-scores
(r=0.406, P<0.001), white and red blood cell counts,
hemoglobin and hematocrit. Higher levels of hemoglobin, hematocrit
and red blood cells were found in adolescents with metabolic
syndrome than in groups without this condition. Conclusion:
Hematological parameters could be important biological markers of
cardiometabolic risk in adolescents with hyperuricemia.
Keywords: Association, Metabolic syndrome,
Platelet count, Risk, Uric acid.
|
|
T
he prevalence of hyperuricemia is increasing in
adults and adolescents [1]. Although hyperuricemia is not included in
the definition of metabolic syndrome, it is observed in 37.2% of
hypertensive and obese adults, and in 90% of adolescents with primary
hypertension [2]. There are a few studies that demonstrate relationships
between hematological parameters and metabolic syndrome. A previous
study suggested that increased erythropoiesis in peripheral blood could
be part of the metabolic syndrome [3]. Uric acid crystals may cause
inflammation through complement activation and induce platelet
activation resulting in higher mean platelet volume. Patients with
increased mean platelet volume are at risk of death due to ischemic
heart disease [4]. An increased platelet count and mean platelet volume
has been reported in hypertensive adolescents, but not in
pre-hypertensive teenagers [5]. Several studies have reported a positive
association between mean platelet volume and blood glucose, blood
pressure, or lipid profile [6] but adequate attention has not been paid
to possible correlation between hematological parameters and serum uric
acid. The aim of this study was to determine if hematological indices
correlate with serum uric acid levels and lipid profile in adolescents
with hyperuricemia.
Methods
The current research is part of a 10-year
retrospective cohort study of patients registered with the Pediatric
Nephrology Department at Medical University of Bialystok, Poland.
Demographic, clinical, and laboratory data were obtained. Hematological
indices (red and white blood cell count, hemoglobin, hematocrit,
platelet count, and mean platelet volume) serum creatinine, urea, uric
acid, total cholesterol, triglycerides, urinalysis, and glomerular
filtration rate (GFR) were assessed.
The inclusion criteria were:
11- to 21-year-old adolescents with serum uric acid >5.5 mg/dL who were
referred to our department in the primary care office because of
elevated casual blood pressure. The exclusion criteria were heart
failure, diabetes mellitus, renal or hepatic dysfunction, hematological
disease, systemic inflammatory conditions, autoimmune diseases,
secondary hypertension, and treatment with medications that affect uric
acid levels and blood pressure values.
The Bioethics Committee of the Medical University of
Bialystok approved the protocol. Bodyweight and height were measured
using a balance beam scale and a wall-mounted stadiometer, respectively.
Body mass index (BMI) was calculated using standard formula. Age- and
gender-specific reference values for BMI were generated by the LMS
method [7]. The LMS values were taken from the study by Kulaga, et
al. [8]. We defined metabolic syndrome according to the criteria
established by Cook, et al. [9]. The diagnosis of hypertension
was based on recommendations of European Society of Hypertension [10],
and hyperuricemia was defined as serum uric acid level >5.5 mg/dL.
Overweight and obesity were defined using criteria developed by Cole,
et al. [10].
Blood samples were collected in the morning, after
overnight fasting. The venous blood samples were combined with
dipotassium EDTA and tested within 30 minutes of collection. Complete
blood counts were measured using an automated blood counter. Serum
creatinine was determined by updated Jaffe reaction and uric acid was
assessed using the colorimetric method. Serum cholesterol,
HDL-cholesterol and triglycerides were determined by the enzymatic
method using Hitachi 912 (La Roche Japan). Serum glucose was measured
with the Integra 800 analyzer. GFR was assessed by updated Schwartz
formula [11].
Results
We examined 607 adolescents (474 boys) during the
study period; 187 (31%) had metabolic syndrome. In the adolescents with
hyperuricemia but without meta-bolic syndrome (420), primary
hypertension was found in 48% and obesity in 19.7% patients. There was
a statistically significant positive correlation between serum uric acid
level and BMI-Z-score (P<0.001), white blood cell count (P<0.001)
and red blood cell count (P<0.001), hemoglobin (P<0.001),
and hematocrit (P<0.001).
The study showed statistically significant
correlations between white blood cell count and lipid profile in
hyperuricemic adolescents. White blood cell count correlated positively
with total cholesterol (r =0.14, P<0.001) and triglycerides (P=0.032),
and negatively with high-density lipoproteins (P=0.045).
Additionally, a positive relationship was found between BMI Z-score and
platelet count (P=0.002) and between BMI Z-score and red blood
cell count (P=0.005), and a negative relationship between BMI
Z-score and mean platelet volume (P=0.038) (Fig. 1).
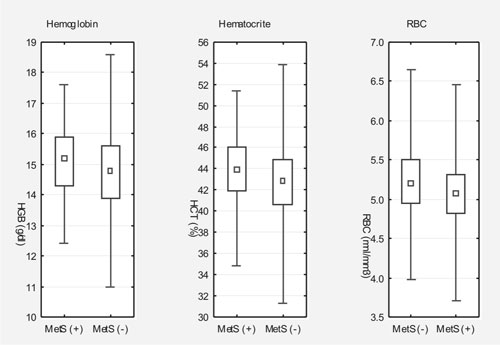 |
|
Fig. 1 Comparison of hematological
parameters between adolescents with (MetS (+)) and without (MetS
(-)) metabolic syndrome.
|
The results showed significantly higher hemoglobin,
hematocrit, and red blood cell count (P<0.001, P<0.001,
and P=0.032, respectively) in patients with metabolic
syndrome than in those without this diagnosis.
Discussion
The results of this retrospective study showed that
in adolescents with hyperuricemia, serum uric acid levels correlated
with white and red blood cell count, hematocrit, and hemoglobin. We
found significantly higher levels of hemoglobin, hematocrit, and red
blood cell count in adolescents with metabolic syndrome than in
the group without it.
Our findings are in general agreement with the
earlier reports [3,12,13] in children and adolescents with obesity,
hypertension, and/or metabolic syndrome. Increased insulin resistance
and other components of the metabolic syndrome were earlier shown to be
associated with increased white and red blood cell count, hemoglobin,
and hematocrit, and as a consequence, increased blood viscosity [14].
Our finding of no correlation between serum uric acid and platelet count
and mean platelet volume was contrary to other reports [15] Although
there is some evidence linking increased white and red blood cell count,
hemoglobin, and hematocrit with hyperuricemia, role of confounding
variables like chronic inflammation and obesity-associated obstructive
sleep apnea cannot be ruled out.
This preliminary study suggests that increased
hematocrit, hemoglobin, and white and red blood cell count could be
important biological markers of metabolic syndrome and hyperuricemia in
adolescents.
Funding: None; Competing interests:
None stated.
|
What This Study Adds?
·
In adolescents with
hyperuricemia, there is a positive correlation between serum
uric acid and white and red blood cell count, hematocrit, and
hemoglobin.
·
Hyperuricemic adolescents with metabolic syndrome – have
higher levels of hemoglobin, hematocrit, and red blood cell
count than those without metabolic syndrome.
|
References
1. Rho YH, Zhu Y, Choi HK. The epidemiology of uric
acid and fructose. Semin Nephrol. 2011;31:410-9.
2. Ahmed N, Anwar W, Huma W. Obesity, hyperlipidemia,
and hyperuraecemia in young and old hypertensive patients. J Ayub Med
Coll Abbottabad. 2009;21:53-6.
3. Kawamoto R, Tabara Y, Kohara K, Miki T, Kusunoki
T, Abe M, et al. Hematological parameters are associated with
metabolic syndrome in Japanese community-dwelling persons. Endocrine.
2013;43:334-41.
4. Slavka G, Perkmann T, Haslacher H, Greisenegger S,
Marsik C, Wagner OF, et al. Mean platelet volume may represent a
predictive parameter for overall vascular mortality and ischemic heart
disease. Arterioscler Thromb Vasc Biol. 2011;31:1215-8.
5. Wasilewska A, Tenderenda E, Taranta-Janusz K,
Zoch-Zwierz W. High-sensitivity C-reactive protein and mean platelet
volume in paediatric hypertension. Pediatr Nephrol. 2010;25:1519-27.
6. Cole TJ, Flegal KM, Nicholls D, Jackson AA. Body
mass index cut offs to define thinness in children and adolescents:
International survey. BMJ. 2007;335:194-204.
7. Kulaga Z, Litwin M, Tkaczyk M, Palczewska I,
Zajaczkowska M, Zwoliñska D, et al. Polish 2010 growth references
for school-aged children and adolescents. Eur J Pediatr.
2010;170:599-609.
8. Cook SS, Weitzman MM, Auinger PP, Nguyen MM, Dietz
WHW. Prevalence of a metabolic syndrome phenotype in adolescents:
Findings from the third National Health and Nutrition Examination
Survey, 1988-1994. Arch Pediatr Adolesc Med. 2003;157:821-7.
9. Lurbe E, Cífková R, Cruickshank JK, Dillon MJ,
Ferreira I, Invitti C, et al. Management of high blood pressure
in children and adolescents: recommendations of the European Society of
Hypertension. J Hypertens. 2009;27:1719-42.
10. Cole TJ, Bellizzi MC, Flegal KM, Dietz WH.
Establishing a standard definition for child overweight and obesity
worldwide: International survey. BMJ. 2000;320:1240-3.
11. Schwartz GJ, Furth SL. Glomerular filtration rate
measurement and estimation in chronic kidney disease. Pediatr Nephrol.
2007;22:1839-48.
12. Pacifico L, Cantisani V, Anania C, Bonaiuto E,
Martino F, Pascone R, et al. Serum uric acid and its association
with metabolic syndrome and carotid atherosclerosis in obese children.
Eur J Endocrinol. 2009;160:45-52.
13. Ford ES, Li C, Cook S, Choi HK. Serum
concentrations of uric acid and the metabolic syndrome among US children
and adolescents. Circulation. 2007;115:2526-32.
14. Brun JF, Aloulou I, Varlet-Marie E.
Hemorheological aspects of the metabolic syndrome: markers of insulin
resistance, obesity or hyperinsulinemia? Clin Hemorheol Microcir.
2004;30:203-9.
15. Shimodaira M, Niwa T, Nakajima K, Kobayashi M,
Hanyu N, Nakayama T. Gender differences in the relationship between
serum uric acid and mean platelet volume in a Japanese general
population. Platelets. 2014;25:202-6.
|
|
|
 |
|

