|
|
|
Indian Pediatr 2012;49:
958-962 |
 |
DNA Damage in Children Exposed to Secondhand
Cigarette Smoke and its Association with Oxidative Stress
|
|
Kabil Shermatov, *Dost Zeyrek, Faruk Yildirim, Mehmet Kilic,
#Nazime Cebi and
†Abdurrahim Kocyigit
From the Harran University Medical School, Department of Pediatrics,
*Harran University Medical School, Department of Pediatrics, Division of
Pediatric Allergy and Pulmonology, #Karadeniz Tecnical University
Medical School, Department of Biochemistry, and †Harran University
Medical School, Department of Biochemistry, Turkey.
Correspondence to: Dr Dost Zeyrek, Harran University School
Medicine, Department of Pediatrics, Division of Allergy and Pulmonology,
TR-63100, Sanliurfa, Turkey. [email protected].
Received: October 05, 2011;
Initial review: October 08, 2011;
Accepted: March 05, 2012.
Published online: 2012, June 30.
PII:S097475591100831-1
|
Objective: To compare oxidative status, total
antioxidant capacity and values of DNA damage in peripheral blood
lymphocytes in children exposed to secondhand cigarette smoke with
healthy controls.
Design: Analytical, Observational.
Participants: 54 children without any chronic
diseases, attending the healthy child monitoring polyclinic. These
comprised 27 children who had been exposed to passive cigarette smoke
and 27 children who had not been exposed to cigarette smoke.
Main Outcome Measures: Urine cotinine levels by
the chemiluminescent technique; DNA damage by alkaline comet assay; and
total oxidant status (TOS) using a novel automated measurement method.
Results: The mean urine cotinine, TOS, Oxidative
Stress Index (OSI) and DNA damage values of the group exposed to
cigarette smoke were determined to be at significantly higher level
compared to the group not exposed to cigarette smoke (P<0.001).
No statistically significant difference was determined in the TAS level
between the two groups (P=0.1)
Conclusions: The results showed that TOS levels,
OSI index and DNA damage in peripheral blood lymphocytes were
significantly higher in children exposed to secondhand cigarette smoke
than in those not exposed to secondhand cigarette smoke.
Key words: Antioxidant status, Children, DNA damage, Oxidant
status, Secondhand cigarette smoke.
|
|
C
igarette smoke inhalation causes cancer in
various organs, and smoking during pregnancy harms both mother and baby,
initially retarding intrauterine development with several side-effects
[1]. Various respiratory diseases can be seen in children, even at
low-level exposure to environmental cigarette smoke [2].
Cigarette smoke contains several free radicals which
may damage lipids, proteins, DNA, carbohydrates and other biomolecules
[3]. Increased production of reactive oxygen species (ROS) leads to an
imbalance between the oxidative forces and the antioxidant defence
systems favoring an oxidative injury.
DNA is a particular target for oxidation as damage
may lead to important alterations. Many oxidative footprints are thought
to be the result of nonenzymatic reactions between reactive oxygen
species and organic molecules, such as proteins, lipids, or DNA. It has
been proposed that DNA damage induced by ROS may contribute to increased
mutation rates, genome instability, apoptosis and associated tissue
regeneration and cell proliferation [4]. Therefore, this study aimed to
compare oxidative status, total antioxidant capacity and values of DNA
damage in peripheral blood lymphocytes in children exposed to secondhand
cigarette smoke with those of healthy controls who had not experienced
secondhand cigarette smoke exposure.
Methods
A total of 54 children who had no chronic diseases
and were attending the healthy child monitoring polyclinic at Harran
University Practice and Research Hospital between July and September
2010 were enrolled into the study. These comprised 27 children who had
been exposed to passive cigarette smoke and 27 children who had not been
exposed to cigarette smoke. Those who had been exposed to environmental
cigarette smoke, although they did not smoke themselves (daily exposure
to at least 1 cigarette or at least 2 hours exposure to environmental
cigarette smoke) and who had a urine cotinine level below 200 ng/mL were
accepted as the passive smoking group and those whose parents did not
smoke and had not been exposed to environmental cigarette smoke and had
a urine cotinine level below 30ng/mL, formed the control group [5].
Approval was obtained from the Local Ethics Committee
for this cross-sectional, controlled study and informed consent was
obtained from the parents of all the children. Data was collected by the
researcher through face-to-face interviews.
Measurement of Urine Cotinine and Creatinine: A
urine sample was taken from each child in a sterile and closed urine
tube. At the same time a 5 cc blood sample was taken into a heparinized
tube for examination of mononuclear leukocyte DNA damage. The assessment
of urine cotinine levels was made by the chemiluminescent technique
using DPC Immulite 2000 (Siemans USA). Cotinine levels were calculated
as ng/ml. Variations may be seen because cotinine expression in the
urine is dependent on the amount of creatinine, so the urine creatinine/cotinine
ratio was calculated. Creatinine measurements were made from spot urine
samples using the Jaffe colormetric technique with the Abbott Architect
C16000 autoanalyser commercial kit (Abbott Laboratories, USA).
After overnight fasting, venous blood was withdrawn
into heparinized tubes and citrated tubes. One mL of heparinized blood
was pipetted into another tube immediately to measure lymphocyte DNA
damage. The remaining blood was centrifuged at 1300 g for 10 min to
separate the plasma. The plasma samples were stored at –80 o
C until analysis of total antioxidant status (TAS) and total oxidant
status (TOS).
Lymphocyte separation: An amount of 1 mL
heparinized blood was carefully layered over 1 mL Lympoprep (Sigma and
Aldrich, Oslo, Norway) and centrifuged for 35 min at 500 g and 25 o
C. The interface band containing lymphocyte was
washed with phosphate-buffered saline (PBS) and then collected by 15 min
centrifugation at 400 g. The resulting pellets were resuspended in PBS.
Membrane integrity was assessed by means of Trypan Blue exclusion
method.
Measurement of lymphocyte DNA damage: The
endogenous lymphocytes DNA damage was analyzed by alkaline comet assay
according to Singh, et al. [6] with minor modifications. Ten mL
of fresh lymphocyte cell suspension (around 20,000 cells) was mixed with
80 mL of 0.7% low-melting-point agarose (LMA) (Sigma) in PBS at 37ºC.
Subsequently, 80µL of this mixture was layered onto slides that had
previously been coated with 1.0% hot (60º C) normal melting point
agarose (NMA), covered with a cover-slip at 4º C for at least 5 min to
allow the agarose to solidify. After removing the cover-slips, the
slides were submersed in freshly prepared cold (4º C) lysing solution
(2.5 M NaCl, 100 mM EDTA-2Na; 10 mM Tris–HCl, pH 10-10.5; 1% Triton
X-100 and 10% DMSO added just before use) for at least 1 hr. Slides were
then immersed in freshly prepared alkaline electrophoresis buffer (0.3
mol/l NaOH and 1 mmol/l Na2ETDA, pH > 13) at 4ºC for unwinding (40 min)
and then electrophoresis is done (25 V/300 mA, 25 min). All of the above
steps were conducted under red light or without direct light in order to
prevent additional DNA damage. After electrophoresis, the slides were
stained with ethidium bromide (2 µ/mL in distilled; 70 µl/slide),
covered with a coverslip and analyzed using a fluorescence microscope
(Nikon, Japan) vided with epi-flourescence and equipped with rhodamine
filter (excitation wavelength, 546 nm; barrier filter, 580 nm) The
images of 100 randomly chosen nuclei (50 cells from each of two
replicate slides) were analyzed visually from each subject, as described
elsewhere. Each image was classified according to the intensity of the
fluorescence in the comet tail and was given a value of either of 0, 1,
2, 3, or 4 (from undamaged class 0 to maximally damaged class 4), so
that the total scores of the slides could be between 0 and 400 arbitrary
units (AU). All procedures were completed by the same biochemistry staff
and DNA damage was detected by a single observer who was not aware of
the subject’s diagnosis.
Plasma TAS levels were determined using a novel
automated measurement method, developed by Erel [7]. Plasma TOS levels
were determined using a novel automated measurement method, developed by
Erel [8].
Statistical analysis: Data were analyzed
using the SPSS for Windows (Version 11.5). All the values are expressed
as mean ± SD. For a comparison of differences between the children
exposed to secondhand cigarette smoke and the control group, chi-squared
test and Student’s t-test or Mann Whitney U-test were used
for non-continuous and continuous variables, respectively. Correlation
analyses were performed using Pearson’s correlation test and Spearman’s
rank correlation. Statistical significance was defined at P<0.05.
Results
A total of 54 children were enrolled in the study,
comprising 27 passive smoking and 27 not exposed to cigarette smoke.
There was no statistical significant difference between the groups in
terms of gender, age, weight, height and body mass index (Table
I).
TABLE I A Comparison of Mean Age, Height, Weight and BMI Values of the Children in the Study
|
Exposed to |
Not exposed to |
P
|
|
cigarette smoke |
cigarette smoke
|
|
|
(n=27) |
(n=27) |
|
|
Age (y) |
5.1 ± 0.8 |
5.4 ± 0.7 |
0.15 |
|
Weight (kg) |
17.7 ± 3.1 |
18.1 ± 2.8 |
0.59 |
|
Height (cm) |
110.5 ± 7.9 |
112.5 ± 7.0 |
0.34 |
|
BMI (kg/m2) |
14.3 ± 1.1 |
14.2 ± 1.3 |
0.77 |
|
Values in mean ± SD; TAS: Total anti-oxidant status; TOS: Total
oxideant status; OSI: Oxidative stress index. |
The mean urine cotinine, TOS, OSI and DNA damage
values of the group exposed to cigarette smoke were at a statistically
significantly high level compared to the group not exposed to cigarette
smoke, but no difference was determined in the TAS level (Table
II). Children exposed to cigarette smoke were allocated into two
groups according to the number of cigarettes exposed to daily; 22
children (85%) were exposed to 1-10 cigarettes per day; 5 children (15%)
were exposed to >10 cigarettes per day. A statistically significant
difference was determined between these two groups in terms of urine
cotinine, TOS, OSI and DNA damage levels but there was no difference in
the TAS levels (Table III).
TABLE II Urine Cotinine, TAS, TOS, OSI and DNA Damage Levels of the Groups
|
Exposed to |
Not exposed to |
P
|
|
cigarette smoke |
cigarette smoke
|
|
|
(n=27) |
(n=27) |
|
|
Cotinine (ng/mL) |
71.55±39.86 |
18.81±13.55 |
<0.001 |
|
TAS (mmolTroEqv/L)
|
0.95±0.16 |
1.02±0.13 |
0.10 |
|
TOS (µmolH2O2Eqv/L) |
32.39±10.19a |
19.61±6.26a |
<0.001 |
|
OSI (AU) |
3.21±1.39a |
1.93±0.67a |
<0.001 |
|
DNA damage (AU) |
62.14±56.31 |
6.14±5.51 |
<0.001 |
|
Values in mean ± SD; TAS: Total anti-oxidant status; TOS: Total
oxidant status; OSI: Oxidative stress index. |
TABLE III Urine Cotinine, TAS, TOS, OSI Values According to the Number of Cigarettes Smoked by Parents
|
1-10 cigarettes/d |
>10 cigarettes/d |
P |
|
(n=22) |
(n=5) |
|
|
Cotinine (ng/mL) |
32.55±20.30 |
66.53±37.26 |
0.005 |
|
TAS (mmolTroEqv/L)
|
0.97±0.16 |
0.96±0.15 |
0.950 |
|
TOS (µmolH2O2Eqv/L)
|
24.27±5.12 |
26.12±7.36 |
0.061 |
|
OSI (AU) |
2.22±0.55 |
3.76±1.16 |
0.009 |
|
DNA damage (AU) |
11.20±7.56 |
75.09±54.96 |
0.01 |
|
Values in mean ± SD; TAS: Total anti-oxidant status; TOS: Total
oxideant status; OSI: Oxidative stress index. |
There was a significant degree of correlation between
the mean cotinine level of the group exposed to cigarette smoke and TOS,
OSI and DNA damage (Fig. 1). A statistically significant
correlation was found between TOS and OSI values and DNA damage of the
group exposed to cigarette smoke (Fig. 2)
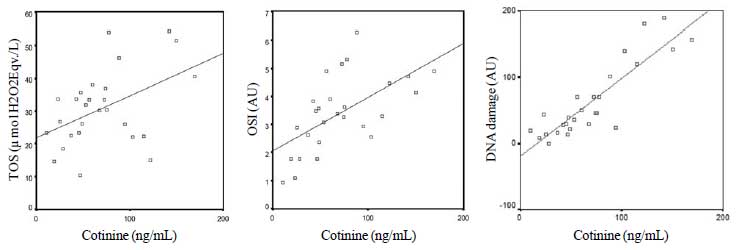 |
|
Fig. 1 Relation between cotinine level and
Total oxidant status TOS (r-0.38); Oxidative
stress index; OSI (r=0.65): and DNA damage (r=0.84) levels in
the group exposed to cigarette smoke.
|
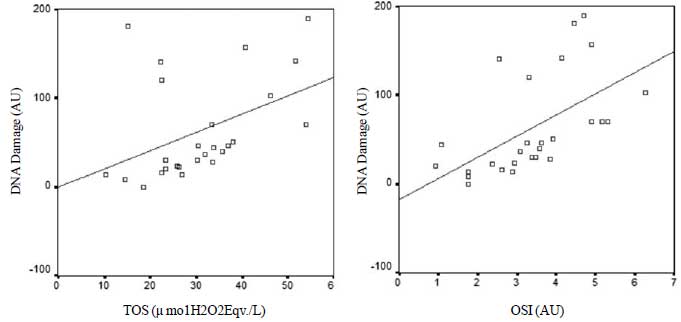 |
|
Fig. 2 Relation between the level of
DNA damage and Total oxidant status TOS (r=0.53); Oxidative
stress index OSI (r=0.72) levels of the group exposed to
cigarette smoke.
|
Discussion
In common with worldwide trends, Turkey is facing the
significant health problem of children exposed to cigarette smoke.
According to the European Tobacco Control Report 2007, the prevalence in
Turkey of the passive effects of cigarette smoke on the 13-15-year old
age group is 81.6% at home and 85.9% outside the home [9].
The harmful effects of direct exposure and passive
smoking have been made known in several studies [10]. A study by
Kocyigit, et al. [11] determined that smoking filter-cigarettes
and hand-rolled cigarettes both strongly increase DNA damage and
oxidative stress in humans. However, both DNA and lipids are more
negatively affected by the smoke from hand-rolled cigarettes. These
findings indicate a correlation between the extent of exposure to
cigarette smoke and DNA damage and OS. Measuring the degree of passive
smoking is of critical importance regarding the carcinogenic effect and
various health problems which occur in children [12].
The results of the present study show that TOS
levels, OSI index and DNA damage in peripheral blood lymphocytes were
significantly higher in children exposed to secondhand cigarette
smoke than in those not exposed to secondhand cigarette smoke. In the
only published study of passive smoking and DNA damage in children [13],
serum MDA concentration as an indicator of oxidative stress and DNA
damage was found to be high in passive smoking children.
In the current study, a significant correlation was
determined between OSI, TOS and DNA damage in children exposed to
cigarette smoke. It has been demonstrated that oxidative stress can lead
to DNA damage, including DNA adducts, strand breaks and other lesions
[14]. In addition, the correlation between OS and DNA damage determined
in various studies indicates that DNA damage is related to OS [15].
However, in the current study, no difference was
determined between the TAS levels of the 2 groups and no correlation was
determined between TAS and DNA damage. Antioxidant level of smokers was
determined to be low in a previous study [13]. It has been proposed that
the development of the antioxidant system following increased OS from
exposure to smoke could be a metabolic self-defence adaptation. Several
published studies have put forward the idea that when the oxidant system
increases, there is a decrease in the antioxidant system [16,17]. In
contrast, a study of asthmatic children by Zeyrek, et al. [15]
determined the TAS level to be high. Nadeem, et al. [16-18]
recorded that when there was an increase in the oxidant system there was
also an increase in the antioxidant system. Host antioxidant systems are
generally activated in response to an oxidant attack, but individuals
have different capacities of antioxidant defence, which are in part
genetically determined [19]. A study by Ercan, et al. [20] showed
that there were genetic differences in the antioxidant response. Various
other studies have shown that as oxidative stress increases, so the
antioxidant capacity increases as a protective mechanism [21].
DNA damage and OSI were determined to be at a
significantly high level in the group exposed to more than 10 cigarettes
per day (although the statistical value was low because the number in
the group was low). Also, a positive correlation was determined between
DNA damage, OSI and cotinine level. The study by Zalata, et al.
[13] determined a statistically significant relationship between the
degree of exposure and DNA damage and oxidative stress. These findings
indicate that the severity of exposure is important.
In conclusion, this study of passive smoking children
has shown DNA damage and OSI by measuring the level of urine cotinine as
an objective criteria of exposure to cigarette smoke. Despite the
findings having been determined by reliable methods, a limitation of the
study is that the number included in the study was low and because there
is widespread exposure to secondhand cigarette smoke in our study
population, the number of control cases was insufficient.
Many studies of adult smokers and passive smokers
have reported various substances in cigarette smoke which show a
genotoxic effect by damaging the cellular DNA structure [22,23]. Various
studies have shown a relationship between cancer and exposure to
cigarette smoke in both adults and children [24]. It is thought that
future studies of varied cohorts aimed at determining the relationship
between DNA damage occuring in children exposed to cigarette smoke and
the development of cancer, will increase the importance of these
findings.
Contributors: All the authors have contributed,
designed and approved the study; Funding: The research was funded
by the Research Fund of Harran University School of Medicine;
Competing interests: None stated.
|
What is Already Known?
•
Exposure to passive smoking in children is reported to cause
DNA damage and increased oxidative stress.
What This Study Adds?
•
We document similar findings in Turkish children.
|
References
1. Florescu A, Ferrence R, Tom T, Selby P, Koren G.
Methods for quantification of exposure to cigarette smoking and
environmental tobacco smoke: Focus on developmental toxicology. Ther
Drug Monit. 2009;31:14-30.
2. De Sario M, Forastiere F, Viegi G, Simoni M,
Chellini E, P. Piccioni P, et al. Parental smoking and
respiratory disorders in childhood, Epidemiol Prevent. 2005;29:52-6.
3. Eiserich JP, van der Vliet A, Handelman GJ,
Halliwell B, Cross CE. Dietary antioxidants and cigarette smoke-induced
biomolecular damage: a complex interaction. Am J Clin Nutr. 1995;62(6
Suppl):1490S-1500S.
4. Sawa T, Ohshima H. Nitrative DNA damage in
inflammation and its possible role in carcinogenesis. Nitric Oxide.
2006:14:91-100.
5. Bramer SL, Kallungal BA. Clinical considerations
in study designs that use cotinine as a biomarker. Biomarkers.
2003;8:187-203.
6. Singh NP, McCoy MT, Tice RR, Schneider EL. A
simple technique for quantitation of low levels of DNA damage in
individual cells. Exp Cell Res. 1988;175:184-91.
7. Erel O. A novel automated direct measurement
method for total antioxidant capacity using a new generation, more
stable ABTS radical cation. Clin Biochem. 2004;37:277-85.
8. Erel O. A new automated colorimetric method for
measuring total oxidant status. Clin Biochem. 2005;38:1103-11.
9. The European Tobacco Control Report 2007. Accessed
Sep4,2011
http://www.euro.who.int/_data/assets/pdf_file/0005/68117/E89842.pdf.
Accessed Sep 4,2011.
10. Jinot J, Bayard S. Respiratory health effects of
exposure to environmental tobacco smoke. Rev Environ Health.
1996;11:89-100.
11. Kocyigit A, Selek S, Celik H, Dikilitas M.
Mononuclear leukocyte DNA damage and oxidative stress: The association
withsmoking of hand-rolled and filter-cigarettes. Mutation Research.
2011;721:136-41.
12. Dabson R. Passive smoking increases children’s
risk of nasal cancer. BMJ. 2005;331: 534-5.
13. Zalata A, Yahia S, El-Bakary A, Elsheikha HM.
Increased DNA damage in children caused by passive smoking as assessed
by comet assay and oxidative stress. Mutat Res. 2007; 629:140-7.
14. Sanders SP, Zweier JL, Harrison SJ, Trush MA,
Rembish SJ, Liu MC. Spontaneous oxygen radical production at sites of
antigen challenge in allergic subjects. Am J Respir Crit Care Med.
1995;151:1725-33.
15. Zeyrek D, Cakmak A, Atas A, Kocyigit A, Erel O.
DNA damage in children with asthma bronchiale and its association with
oxidative and antioxidative measurements. Pediatr Allergy Immunol.
2009;20:370-6.
16. Liao MF, Chen CC, Hsu MH. Evaluation of the serum
antioxidant status in asthmatic children. Acta Paediatr Taiwan.
2004;45:213-7.
17. Hanta I, Kuleci S, Canacankatan N, Kocabas A. The
oxidant-antioxidant balance in mild asthmatic patients. Lung.
2003;181:347-52.
18. Nadeem A, Chhabra SK, Masood A, Ral HG. Increased
oxidative stress and altered levels of antioxidants in asthma. J Allergy
Clin Immunol. 2003;111:72-8.
19. Barnes PJ. Reactive oxygen species and airway
inflammation. Free Radic Biol Med. 1990; 9:235-43.
20. Ercan H, Birben E, Dizdar EA, Keskin O, Karaaslan
C, Soyer OU, et al. Oxidative stres and genetic and epidemiologic
determinants of oxidant injury in childhood asthma. J Allergy Clin
Immunol. 2006;118:1097-104.
21. Sen CK. Oxidants and antioxidants in exercise. J
Appl Physiol. 1995;79:675-86.
22. Palma S, Cornetta T, Padua L, Cozzi R, Appolloni
M, Ievoli E, et al. Influence of glutathione S-transferase
polymorphisms on genotoxic effects induced by tobacco smoke. Mutat Res.
2007;633:1-12.
23. Sasikala K, Rosalin FR, Jude ALC, Kumar RA, Sudha
S, Devi MV, et al. Active and passive smokers - a
haematobiochemical and cytogenetic study. Int J Hum Genet. 2003;3:29-32.
24. Wang FL, Love EJ, Lin N, Dai XD. Childhood and
adolescent passive smoking and risk of famale lung cancer. Ýnt J
Epidemiol. 1994;23:223-30.
|
|
|
 |
|

