|
|
|
Indian Pediatr 2010;47: 679-686 |
 |
Locally Made Ready-to-Use Therapeutic Food for
Treatment of Malnutrition:
A Randomized Controlled Trial |
|
Azara Sneha Singh,
*# Gagandeep
Kang,* Anup Ramachandran,* Rajiv Sarkar,* Pearline Peter#
and Anuradha Bose#
From the Departments of *Gastrointestinal Sciences and
# Community Health, Christian
Medical College,
Vellore TN 632004, India.
Correspondence to: Anuradha Bose, Professor of
Pediatrics, Department of Community Health, Christian Medical College,
Vellore, TN, 632002 India.
Email: [email protected]
|
|
Abstract
Objective: To evaluate the effectiveness of a
locally made ready-to-use therapeutic food (RUTF) in decreasing mild to
moderate malnutrition.
Design: A randomized open label, controlled
trial.
Setting: Pre-schools run by the Department of
Community Health in Kaniyambadi administrative block, Vellore, India;
duration of follow-up – 3 months from the date of recruitment.
Participants: Pupils aged 18-60 months with
Weight-for-Age £2 SD.
Interventions: A locally produced energy-dense
supplement (RUTF), and the current standard of care [teaching caregivers
how to make a fortified cereal-milk supplement called High Calorie
Cereal Milk (HCCM)].
Main outcome measures: Increase in
weight-for-age status; increase in levels of plasma zinc, vitamin B12,
serum albumin and haemoglobin.
Results: The Mean (SD) weight gain at 3 months
was higher in the RUTF group: RUTF (n=51): 0.54 kg; (SE = 0.05;
95% CI = 0.44 – 0.65) vs HCCM (n=45): 0.38 kg; (SE = 0.06;
95% CI = 0.25 – 0.51), P = 0.047. The weight gain per kilogram of
body weight was directly proportional to the severity of malnutrition.
Conclusions: Community-based treatment showed
weight gain in both groups, the gain being higher with RUTF.
Key words: India, Indigenous, Management,
Malnutrition, Nutrition therapy.
Registered at the Clinical Trials Registry of India; Registration
number: CTRI/2009/09/000007.
|
|
T
he third National Family Health
Survey estimated that 45.9% of Indian children and 33.2% of children in
Tamil Nadu below 3 years of age are underweight(1). Mild-to-moderate
malnutrition has been associated with an increased risk of childhood
mortality(2,3). Treatment of malnutrition has largely been restricted to
health education directed at increasing the caloric and protein content of
a young child’s diet. Severe malnutrition has been treated in a hospital
setting.
Ready to use high energy foods to be provided to the
caregiver of a malnourished child has been a paradigm shift in the
management of malnutrition. Evidence is largely restricted to severe
malnutrition, where home-based and community-based therapy with
nutrient-dense foods has been found to be more cost-effective than
in-patient care of severe acute malnutrition (SAM). Home-based treatment
has been recommended during the rehabilitation phase of treatment for
malnutrition in areas where follow up is possible(4). The use of
ready-to-use therapeutic foods (RUTF) for the treatment of moderate
malnutrition has been reported to result in an average weight gain of
12.7% over a period of 28 days in moderately malnourished children(5).
This trial was designed to evaluate the effectiveness
of community-based therapy with a locally produced RUTF in comparison with
the standard of care, for treatment of all grades of malnutrition, by
anthro-pometry, measurement of micronutrient levels, and intestinal
function in children < 60 months attending local pre- schools.
Methods
This study was an open-labeled randomized controlled
trial. Children were randomly assigned to either receive a locally
produced energy-dense RUTF, administered in pre-schools by teachers,
compared to the current standard of care: to teach caregivers to prepare a
fortified cereal-milk supplement (High-Calorie Cereal Milk; HCCM), and
advised 2 x 100 ml feeds per day. Both arms included continuation of
family diets.
Sample size estimation
A total of 120 children (60 in each intervention
group), assuming a 5% drop-out had 80% power to detect a difference of 50%
of reduction in the proportion of malnutrition between groups, with an
overall type I error of 5%.
334 children between the ages of 18 to 59 months were
screened in 16 pre-schools, information leaflets sent home with the
parents, the parents met at a later date, and 128 children recruited after
obtaining written informed consent. Ten were later excluded. The study was
approved by the Institutional Review Board of the Christian Medical
College, Vellore, India.
Block randomization was done in blocks of ten using a
computer-generated sequence, generated by the statistician. The children
were allocated to either group by one of the investigators. Measurement of
anthropometry, measurement of micronutrient levels and intestinal function
were done.
Participants in the study were children recruited from
16 village pre-schools run by the Community Health and Development (CHAD)
Hospital of the Christian Medical College, Vellore. Children aged 18-60
months, -2 SD weight-for-age and below but not requiring hospitalization
for malnutrition, were considered eligible. Children younger than 18
months were excluded as several of them were receiving a predominantly
milk diet, as chosen by their parents.
The children received supplementation for three months
from recruitment, the study running from January to the end of May 2008.
Baseline assessment of nutritional status, micronutrient levels and
intestinal function were carried out immediately following recruitment.
Weight and height were measured, and blood samples taken for estimation of
serum albumin, plasma zinc, plasma vitamin B12, hemoglobin and red cell
indices. The micronutrients chosen for assay were those that were
considered clinically useful. All children underwent D-Xylose testing for
evaluation of intestinal function.
Weight was measured to the nearest 100g on a regularly
calibrated electronic scale with the child standing barefoot and
undressed. Height was measured to the nearest 1 mm using standard
measuring techniques, the mean of two readings was calculated for each
child. Weight and height at day 30, day 60, and day 90 after recruitment,
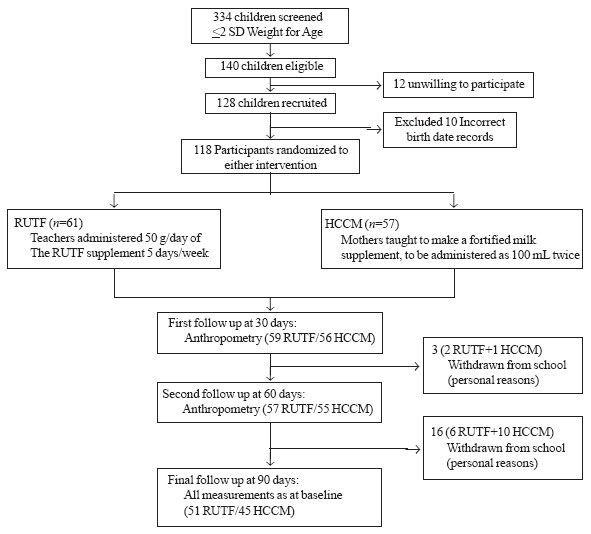
with a window of +5 days to allow for holidays and weekends (Fig.1).
All measurements were done by one investigator. Blood were drawn by a
research nurse. Definition of anemia was as defined by the World Health
Organization for children aged 6 months to 5 years (Hb<11 g/dL)(8). Five
mL of venous blood was drawn, at recruitment and final follow-up. The
tests for hemoglobin and red cell indices were conducted using the Sysmex
KX21 auto analyzer. Plasma zinc was tested using an atomic absorption
spectrometer (Perkin Elmer AAnalyst200). Serum albumin was tested by a
colorimetric method using bromocresol green as an indicator on an
Olympus/Hitachi 912 auto analyzer. Vitamin B 12
was tested using the Roche Immunoassay system. Five-hour urine samples
were collected after the oral administration of D-Xylose according to the
child’s weight, to measure intestinal barrier function. This was done at
recruitment and final follow-up. Urine D-Xylose testing was estimated by
colorimetry using Phloroglucinol(6). Body impedance anthropometry was done
on day 60 and day 90 following recruitment. Baseline measurements are not
available.
 |
|
Fig. 1 Study Flow Chart. |
The primary outcome of the study was recovery, defined
as the attainment of a Weight-for-Age Z score >-2. Secondary outcomes
were changes in the vitamin B 12,
plasma Zinc, serum albumin levels and iron status of the children.
Dietary Interventions
The RUTF was prepared weekly, under supervision, at a
local bakery and packed into new polythene bags (250 g per bag),
heat-sealed and distributed to the pre-schools. The RUTF was produced by
mixing together ground roasted peanut powder, milk powder, and sugar in a
ratio of 30:28:25 (grams), along with 15 grams of gingili oil.
Multivitamin supplements of 2 grams to the above ratio of mix resulted in
a medicinal after-taste after a week, which was unacceptable to the
children and this quantity was reduced to 1 tablet per 100 g of mix. Oral
multi-vitamins were supplemented. The teachers were weekly provided with
one 250 g bag of RUTF for each child, to be administered at a rate of 50 g
per child per working day, at 5.5 Calories per Gram. The RUTF was given
during the mid morning and mid afternoon breaks, approximately 50 g per
working day, equal to about 1.5 tablespoons per helping.
Mothers of the children receiving, High Caloric Cereal
Milk (HCCM) were taught how to make the supplement. HCCM consisted of 100
mL milk fortified with 15 g flour of mother’s choice, 5 mL oil and 2
teaspoons of sugar, cooked to a porridge-like consistency. Two servings of
HCCM made with 100 mL of milk each, were advised, and were to be given at
home.
All children additionally continued to receive their
normal diets, including one hot meal provided by the preschool every
working day as part of the PTMGR Nutritious Meal Program, where each child
below six years of age receives a nutritious noon meal prepared with rice,
dhal, oil and vegetables containing a caloric value of 358.2 to 780.3 and
8.62 g to 12.55 g of protein served every school day(7). The nutritive
values of the 2 products are dissimilar, RUTF containing 550 Cal per 100 g
of product and HCCM containing 187 Calories per 100 mL.
Recruitment was done in January and the intervention
started from the date of recruitment. Measurements were taken on day 1,
day 30, 60 and 90, with a window of 5 days after the scheduled date,
allowing for holidays and weekends. The last measurement of the children
recruited last was completed at the end of May 2008.
Anthropometric indices were calculated using WHO Anthro
(v 2.0.2; Department of Nutrition, World Health Organization). To compare
the differences between the two groups, the chi square test was performed
for dichotomous outcomes and one-way ANOVA or t-test for continuous
outcomes. Weight gain in the two groups was compared using the linear
regression analysis. All analysis were performed using SPSS version 15.0
and Epiinfo version 2002.
Results
Of the 140 malnourished children identified, 128 were
recruited (Fig 1). Confirmation of dates of birth and
rechecking weight resulted in 10 children being excluded. 118 children
were enrolled (Table I). At the end of the study, 96
children remained for follow-up, of whom 51 received RUTF and 45 received
HCCM. Mean (SD) weight gain in 3 months was as follows: RUTF (n=51):
0.54 kg; (SE = 0.05; 95% CI = 0.44 – 0.65) and in the group that received
HCCM (n=45): 0.38 kg; (SE = 0.06; 95% CI = 0.25 – 0.51). The linear
regression analysis showed that the group receiving RUTF gained 0.168 kg
(95% CI 0.002 – 0.333; P=0.046) more than the other group. The data
for the individual children in the group were aggregated. Table
II shows the weight gain (g)/kg body weight/day during the subsequent
month for the different weight-for-age Z score classifications in the
children in the two groups daily categorized according to the degree of
malnutrition. The table shows that the greater the degree of
malnutrition, the higher the weight gain; with a seemingly higher weight
gain in the RUTF group, though the results are not statistically
significant. The gain in weight persisted in the third month only in the
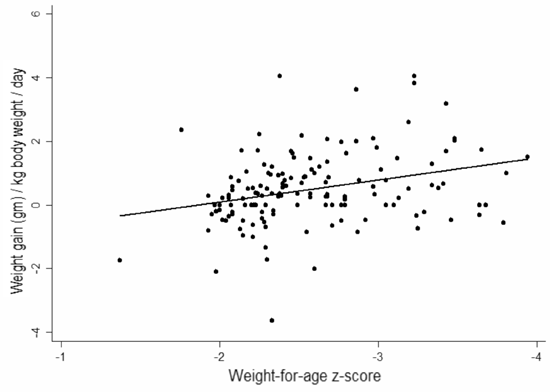
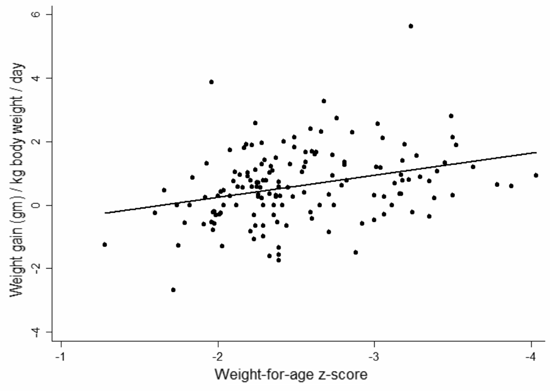
children who were <-3 SD weight for age. Figure 2 shows
scatter plots of the weight gain per kilogram of body weight per day of
the groups receiving the two supplements. In both groups it is evident
that the weight gain per kilogram is higher, the greater the degree of
malnutrition.
TABLE I
Baseline Data for All Children (n=118) Who Completed the Study and Those Who Dropped Out,
Shown Separately
| |
Intervention |
|
Characteristics |
Ready-to-use Food
(RUTF) |
High Calorie Cereal
Milk(HCCM) |
| |
Participants |
Dropouts |
Participants |
Dropouts |
| |
(n=61) |
(n=10) |
(n=51) |
(n=12) |
| Gender: Male |
29 (47.5%) |
5 (50%) |
26 (45.6%) |
5 (41.7%) |
| Mean (SD) age (in years) at recruitment |
3.53 (0.87) |
3.61 (0.77) |
3.59 (0.85) |
3.50 (1.06) |
| Median (IQR) number of siblings (n=108) |
1 ( 1 – 2) |
1 (1 – 1) |
1 ( 1 – 2) |
2 (1 – 2.5) |
| Maternal education > Grade V (n=110) |
35 (57.4%) |
8 (80%) |
29 (50.9%) |
5 (41.7%) |
| Mean (SD) weight (in kg) at recruitment |
10.79 (1.28) |
11.07 (1.37) |
10.98 (1.34) |
10.93 (1.58) |
Table II
Mean (SD) Weight Gain (g)/kg Body Weight/Day for the Different Weight-for-age Z Score
Classifications in the Two Groups
|
|
|
|
Intervention |
|
|
|
Weight-for-age |
RUTF* |
High Calorie Cereal Milk* |
|
Z Score |
Month 1 |
Month 2 |
Month 3 |
Month 1 |
Month 2 |
Month 3 |
|
< 0 to –1 |
– |
– |
– |
– |
– |
– |
|
< –1 to –2 |
– |
0.53 (1.40) |
–0.90 (0.93) |
– |
0.40 (1.37) |
–1.02 (1.02) |
|
< –2 to –3 |
1.00 (0.91) |
0.61 (1.05) |
–0.07 (0.90) |
0.65 (1.12) |
0.51 (0.93) |
–0.09 (0.95) |
|
< –3 to –4 |
1.54 (1.42) |
0.50 (0.90) |
0.86 (0.64) |
0.74 (1.06) |
1.61 (1.54) |
0.24 (0.62) |
|
< –4 |
0.92 |
– |
– |
– |
– |
– |
|
* Differences not
statistically significant. |
Significant increases in serum albumin was seen in both
the RUTF [P=0.045] and the HCCM [P=0.027] groups, as were
changes related to anemia, with improvement in both arms, the results of
which are shown in Table III. D-Xylose absorption test
showed improvement of intestinal function. (Table III) At
baseline, 40 (75.5%) children in the RUTF and 42 (79.2 %) in the HCCM
group had abnormal function, while by the end of the study, 27 (57.4%) had
abnormal absorption in the RUTF group and 23 (50%) in the HCCM group.
There were no adverse events reported.
Table III
Change in Micronutrient Status and Urine D-xylose Excretion % of Children After
Three Months of Supplementation
| |
Vitamin B12 |
Plasma zinc |
Albumin |
Hemoglobin |
D-Xylose excretion % |
| |
RUTF |
HCC M |
RUTF |
HCCM |
RUTF |
HCCM |
RUT F |
HCC M |
RUTF |
HCCM |
| Mean |
-70.23 |
-9.64 |
21.76 |
18.67 |
-0.98 |
-0.109 |
-0.447 |
-1.295 |
4.725† |
8.633† |
| 95% CI of the |
-135.74 |
-49.22 |
21.76 |
10.16 |
-0.98 |
0.204 |
-0.447 |
-4.247 |
0.517 |
4.227 |
| difference* |
to |
to |
to |
to |
to |
to |
to |
to |
to |
to |
| |
-4.72 |
29.94 |
11.47 |
27.17 |
-0.193 |
-0.13 |
-0.899 |
1.657 |
8.933 |
13.039 |
| Significance |
0.036# |
0.626 |
0# |
0# |
0.045# |
0.027# |
0.053 |
0.381 |
0.029# |
0# |
*Difference calculated as baseline – final, therefore, positive values of the mean imply a decrease and negative
values an increase in serum levels of the substance measuredF; †decreasing D-Xylose excretion % indicates
improvement in intestinal barrier function: #statistically significant.
|
The cost of one month’s supply of RUTF, at the rate of
250 g per week, or 50 g per week day, for each child was calculated to be
approximately INR 135 (USD 2.95).
 |
|
(a) |
|
(b)
|
|
Fig. 2 Scatter plot showing the weight
gain against the weight for age Z scores for the group receiving (a)
high calorie cereal milk; and (b) the ready to use therapeutic food
supplement. |
Discussion
The RUTF in this study differed from the formulation
recommended by the WHO in the micronutrient composition(9). There was
significant improvement in the macro - and micro-nutritional status of
participants. As the study was done in the time of year that included
school holidays, with children moving on subsequently to other schools, 22
children did not complete 3 months of follow up. Nevertheless, the results
expressed as weight gain per kilogram of body weight per day show that
there is an increase in weight in both arms, with the degree of weight
gain being directly proportional to the severity of the malnutrition. In
the third month, there was no weight gain in the better nourished
children, though the more severely malnourished continued to gain weight.
It appears that RUTF results in a higher weight gain in the first month.
The results are not statistically significant, but this may be worth
pursuing in subsequent studies with larger numbers. It is of note that
both forms of therapy result in weight gain.
The effects of the supplements on zinc are noteworthy,
and though the explanation of the reduction of levels of zinc in both arms
is unclear, it is possible that nutrient interactions with other
micronutrients in the supplement, the presence of phytates or dietary
fiber may have affected its absorption and bioavailability(11,12). It
could perhaps be that the dose given was insufficient to cope with the
increased demand for zinc with the increase in weight.
The WHO recommends treatment of uncomplicated
severe acute malnutrition at home(9). The Indian Academy of Pediatrics (IAP)
has made recommendations for recognition of those children who must be
referred to a facility for treatment(13). The success of home-based
treatment of severe malnutrition would require the provision of a
nutrient-dense supplement which can be safely stored and administered
without much preparation by the caregiver. Commercially available nutrient
dense foods are expensive, and locally produced nutritious mixes have not
been compared to the present standard of care for home based treatment.
The RUTF used in this study was prepared from locally
available ingredients and in a local bakery. The RUTF, or local
modifications, can be prepared safely and in small or large quantities. A
lower incidence of peanut allergies has been noted among children in
developing countries, as compared to those in developed countries,
especially as they may additionally suffer from severe malnutrition and
co-morbid conditions that further suppress immune function(14). However,
this possibility must be considered whenever a peanut based product is
used. In an Indian setting, the community-based treatment of malnutrition
can find extensive application, and the involvement of pre-school teachers
and mothers’ groups in its delivery is a viable option. Any sustainable
treatment for malnutrition has to rely on modifications of local foods.
RUTF itself is used in the acute phase of rehabilitation and is prescribed
as a therapeutic item, not a food.
The study had several limitations. The numbers
available for analysis at the end of the study were small. The study could
have been done in the early part of the academic year, i.e. June to
December, and the movement of children into other schools at the end of
the school year should have been anticipated. The study is not ideal in
that it was not blinded, but blinding would have been difficult for two
very different but acceptable interventions.
To summarize, this study showed that community based
treatment of malnutrition is a feasible, effective and well-accepted
intervention. The ingredients for the RUTF are widely available and the
supplement can be locally produced. The cost-effectiveness of this method
of therapy needs to be evaluated.
Acknowledgments
Dr Mini Jacob, Mrs Margaret and Mrs Charlet, Dr Vinohar
Balraj, Dr Selvakumar, Dr Victoria Job Daniel and Dr Geetanjali Arulappan,
Mrs Manoranjitham, Mr Suresh, Mr Saravanan, Mr Neel Mahesh Naik, Mr Balaji,
Mr Perumal, Mr Vivek and the staff at Jayanthi Bakery, Mr Jayakumar, Ms
Sheela, Ms Parmeshwari, Mr Singarayan, Mr Bhaskar, Mr Satish, Mr
Thyagarajan, Dr Raja Ivan Singh and Dr Kalyani Singh.
Contributors: AB: conceived and designed the study
with GK and both revised the manuscript for important intellectual
content. AB will act as guarantor of the study. AS: conducted the study,
collected data and drafted the paper; AR: conducted the laboratory tests,
and interpreted them; P analyzed the data with A and helped in manuscript
writing. All authors approved the final manuscript.
Funding: The Fogarty International Clinical
Research Scholars Program and the Department of Gastrointestinal Sciences,
Christian Medical College, Vellore. Neither funding body has a conflict of
interest with the study.
Competing interests: None stated.
|
What is Already Known?
• For the treatment of malnutrition, home-based
and community-based therapy with nutrient-dense foods such as RUTF
is more cost-effective
What This Study Adds?
• Using locally made RUTF in the community-based
treatment of childhood malnutrition is feasible and effective in a
rural South Indian setting.
|
References
1. Ministry of Health and Family Welfare, Government of
India. 2005-2006 National Family Health Survey (NFHS-3); 2009.
2. Pelletier DL, Frongillo EA, Jr., Schroeder DG,
Habicht JP. The effects of malnutrition on child mortality in developing
countries. Bull World Health Organ 1995; 73: 443-448.
3. Pelletier DL. The potentiating effects of
malnutrition on child mortality: epidemiologic evidence and policy
implications. Nutr Rev 1994; 52: 409-415.
4. Ashworth A. Efficacy and effectiveness of
community-based treatment of severe malnutrition. Food Nutr Bull 2006; 27:
S24-S48.
5. Ciliberto MA, Sandige H, Ndekha MJ, et al.
Comparison of home-based therapy with ready-to-use therapeutic food with
standard therapy in the treatment of malnourished Malawian children: a
controlled, clinical effectiveness trial. Am J Clin Nutr 2005; 81:
864-870.
6. Eberts T, Sample R, Glick M, Ellis G. A simplified,
colorimetric micromethod for xylose in serum or urine, with phloroglucinol.
Clinical Chemistry 2009; 25: 1440-1443.
7. State Planning Commission, Government of
Tamil Nadu. Annual Plan 2003-04 - Food Security and Nutrition. Available
from: URL: http://www.tn.gov/spc/annualplan/chapter9-11. Accessed on 3
July, 2009.
8. DeMaeyer EM, Dallman P, Gurney JM, Hallberg L, Sood
SK, Srikantia SG. Preventing and controlling iron deficiency anaemia
through primary health care. Geneva: World Health Organization; 1989. p.
26. Available from: URL: http://www.who.int/nutrition/publications/micronutrients/anaemia_iron_deficiency/9241542497/en/.
Accessed on 3 July, 2009.
9. World Health Organization/World Food Programme/United
Nations System Standing Committee on Nutrition/The United Nations
Children’s Fund, 2007. Community-based management of severe acute
malnutrition. A Joint Statement by the World Health Organization, the
World Food Programme, the United Nations Standing Committee on Nutrition
and the United Nations Children’s Fund. Available from: URL: http://www.who.int/nutrition/topics/statement_commbased_malnutrition/en/index.html.
Accessed on 3 July, 2009.
10. Diop el HI, Dossou NI, Ndour MM, Briend A, Wade S.
Comparison of the efficacy of a solid ready-to-use food and a liquid,
milk-based diet for the rehabilitation of severely malnourished children:
a randomized trial. Am J Clin Nutr 2003; 78: 302-307.
11. Sandstrom B. Micronutrient interactions: effects on
absorption and bioavailability. Br J Nutr 2001; 85 Suppl 2 :S181-S185.
12. Greger JL. Nondigestible carbohydrates and mineral
bioavailability. J Nutr 1999; 129: 1434S-5S.
13. Gupta P, Shah D, Sachdev HPS, Kapil U. National
Workshop on Development of guidelines for effective home based care and
treatment of children suffering from severe acute malnutrition. Indian
Pediatr 2006; 43: 131-139.
14. Abbassy AS, el-Din MK, Hassan AI, Arif GH, Hammad
SA, el-Araby II, et al. Studies of cell-mediated immunity and
allergy in protein energy malnutrition. II. Immediate hyper-sensitivity. J
Trop Med Hyg 2009; 1974: 18-21.
|
|
|
 |
|

