Generalized resistance to thyroid hormones (GRTH) is an inherited
syndrome of reduced target tissue sensitivity to thyroid hormones(1,2).
Thyroid hormone resistance was first described in 1967 by Refetoff,
Dewind, and De Groot in several siblings in a family with congenital
deafness and dysgenetic stippled epiphyses; these siblings were
evaluated with clinical suspicion of hypothyroidism but, surprisingly,
their thyroid hormone levels were found markedly elevated(3). Because
there were no signs or symptoms of thyrotoxicosis, the authors
hypothesized the existence of target organ resistance to thyroid hormone
to explain the clinical and biochemical findings. In this report we
describe a family with generalized. thyroid hormone resistance with
variable clinical picture in different siblings.
Case Report
This study was conducted on a couple with 3 children, namely,
A - 9
year male child, B
-
5 year female child and C - one year female child. It was the second
child B who was first brought to medical attention. She was referred to
our endocrine clinic for evaluation of a small swelling in the neck,
short stature, and high normal serum total triiodothyronine(T3),
elevated total thyroxine (T4) and thyroid stimulating hormone
(TSH) by a general practitioner who l1ad failed to interpret the hormone
assay. In our clinic, we received a 5 year old female child, product of
a consanguineous marriage, whose parents were primarily concerned about
her short stature. She was second in birth order born by normal delivery
of a full term pregnancy with uneventful neonatal history. At the age of
4 years, parents had noticed a small swelling in the front of neck and
delay in her linear growth as compared to other sibs. She had no history
of constipation, 'prolonged diarrhea, or vomiting. Clinical examination
revealed a female child with pulse of 60 beats/minute, palpable thyroid
gland (Grade II), without any features of rickets or malnutrition. Her
anthropometric parameters are given in Table I. The systemic
examination was unremarkable.
The investigations performed included complete hemogram, detailed urine
and stool analysis, detailed serum biochemistry including blood glucose,
blood urea nitrogen
(BUN),
creatinine, bilirubin, aspartate aminotransferase (AST) alanine
amionstransferase (ALT), calcium, phosphorus, and alkaline
phosophataseand complete lipidogram. Radiology including X-ray skull,
X-ray chest, and X-ray left wrist for bone age was performed.
In view of an available thyroid function test report with high normal
total T3 and elevated total T4 and TSH and a
possibility of thyroid hormone resistance, the parents and other
siblings of the index case were also evaluated. Though being
asymptomatic, they were explained about the possible genetic nature of
this disorder and were evaluated with consent. All the above mentioned
investigations were performed in the father, mother and two siblings of
the index case. Fig. 1 shows the index case with two siblings.
TABLE I
Anthropometric Parameters and Thyroid Function Tests in the Subjects
Studied
|
Cases |
Chrono-
logic age
(yr) |
Height
(cm) |
Height
age (yr) |
Bone
age (yr) |
Total T3
Normal
value:
0.070-2.50
ng/ml |
Total T4
Normal
value:
5.50-13.50
µg/dl |
TSH
Normal
value:
00.50-6.50
'µIU/ml) |
|
A |
9 |
124 |
8 |
7 |
2.82 |
14.14 |
1.20 |
|
B |
5 |
98 |
3 |
3 |
i.* 1.82 |
18.62 |
2.14 |
|
|
|
|
|
|
i.* 1.82 |
16.67 |
2.14 |
|
|
|
|
|
|
iii.* 2.03 |
20.12 |
2.18 |
|
C |
1 |
75 |
1 |
1 |
2.64 |
15.28 |
1.81 |
|
D |
32 |
176 |
- |
-
|
2.16
|
18.00 |
0.79 |
|
E |
28 |
158 |
-
|
-
|
1.34 |
9.00 |
1.09 |
* T3, T4 and TSH done on three
occasions in the index case.
A: Elder brother B: Index case C: Younger sister D: Father E.
Mother
Hormone estimations done included total T3, total T4
and TSH (thrice in the index case) in the parents and the 2 siblings,
and insulin tolerance test in the index case only. Insulin tolerance
test was performed by giving 0.1 units/kg body weight of regular insulin
intravenously and collecting samples for growth hormone and cortisol
estimation at 0, 30, 60, 90 and 120 minutes(4).
All hormone estimations were done by sensitive radioimmunoassay; T3,
T4, TSH, and growth hormone estimation was performed with
radioimmunoassay kits supplied by Bharat Radioactive Isotope Technology,
Mumbai, India, while cortisol estimation was done by commercially
available kits from Diagnostic Products Corporation, Los Angeles, USA.
The details of anthropometric parameters of the family are given in
Table 1. The baseline investigations-complete blood counts, ESR,
biochemical parameters including blood glucose, urea nitrogen,
creatinine, bilirubin, ALT, AST, calcium, phosphorus and alkaline
phosphatase, and urine and stool analysis were within the normal range
in all subjects. Only the index case had hypercholesterolemia (serum
cholestrol 224 mg/dl). Roentgenogram of skull and chest also were
normal. The bone age of the index case was 3 years.
The details of total T3
total T4
and TSH are given in Table I. The peak growth hormone and
cortisol after insulin induced hypoglycemia in the index case were 20.4
mg/ dl and 30.2
µg/dl,
respectively.
Discussion
In 1942, Fuller Albright used the example of pseudohypoparathyroidism to
introduce the concept of hormone resistance syndrome(5). Over the last
half a century, clinical, biochemical and molecular studies of hormone
resistance syndromes have flourished. The prevalence of thyroid hormone
resistance and heterogeneity
of its
clinical presentation are now becoming apparent, often resulting in
varied and confusing clinical profiles, perhaps due to variable tissue
sensitivity(2,6). Generalized resistance to thyroid hormones is a
syndrome characterized by elevated 'serum free T3 free T4
and TSH levels and preservation of TSH response to thyroid hormone
releasing hormone (TRH) in the absence of clinical manifestations of
thyroid hormone excess(2,7,8). Some patients have severe pituitary as
well as peripheral thyroid hormone resistance accompanied by signs and
symptoms of hypothyroidism(3,8). More commonly, individuals have partial
pituitary and peripheral resistance to thyroid hormones and are
clinically euthyroid(3,9,10). Occasionally, resistance at the level of
pituitary only has been reported; these individuals have symptoms of
hyperthyroidism(1,11,12). Rarely selective peripheral resistance without
pituitary resistance to thyroid hormones has been re- ported; these
patients are clinically
hypothyroid and have normal T
3
T4
and
TSH levels(13).
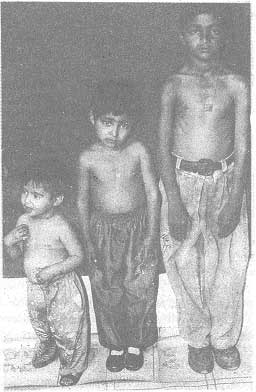 |
Fig. 1. Photograph of the index case with two siblings. |
The clinical features of GRTH can include attention deficit disorders, delayed
skeletal maturation, tachycardia, a variety of somatic abnormalities,
and altered metabolic response to thyroid hormones(2,14). Some individuals exhibit a curious combination
of physical findings such as delayed skeletal maturation (suggestive of
hypothyroidism) and tachycardia, (suggestive of hyperthyroidism), leading to the notion that the degree of hormone resistance varies in different organs(15).
Our index patient had a goiter and some features of hypothyroidism in the form of short stature, bradycardia,
and delayed bone age with high normal total T3, elevated total T4 and normal levels of TSH (reproducible on three separate samples). In view of these findings we believe she had sufficient clinical and biochemical suggestion of GRTH. Elevated
T3 and T4 could also occur in the presence of excessive(16) or abnormal thyroid binding proteins(17,18) but, in view of features of hypothyroidism (short stature, bradycardia and delayed bone age with palpable goiter), without any other overt cause of these findings, it appears logical to entertain a diagnosis of
GRTH.
On evaluation of rest of the family, elevated thyroid hormones were also found in father, brother, and sister of the index case (Table 1). This suggests an autosomal dominant pattern of inheritance of the disorder in this family. The inheritance pattern in most of the earlier reported families with GRTH is autosomal dominant(6,8,9,10). An autosomal recessive type
of inheritance has been reported in only one family(19). Although, cases of sporadic occurrence have been suspected(13,20), it is probable that additional family members might have been found to be affected if studied carefully.
It has long been suspected that GRTH is caused by abnormalities in the human thyroid hormone receptor (hTRβ)(21). However, this hypothesis was confirmed only recently by the identification of mutations in . the hTRβ gene in patients with GRTH(22,23). These mutations alter single aminoacids located in the hormone-binding domain of the hTRβ,
resulting in the expression of receptor proteins with either reduced(24) or no(25,26)
T3 binding activity.
In individuals with GRTH who do not have goiter (as subjects A, C and D), thyroid hormone production is probably adequate to supply the peripheral tissues. This explains their adequate height age and bone age. The development of a goiter or symptoms of hypothyroidism in these individuals would suggest that the gland is not able to adequately compensate for the resistance to thyroid hormones.
It is suggested that every child who presents with hyperthyroxinemia should have
a serum TSH estimation done and, if it is
not suppressed, should be evaluated to exclude thyroid hormone resistance before anti thyroid therapy is initiated. Once the diagnosis has been established, screening of other family members should be under- taken. Increased recognition and evaluation of patients with thyroid hormone resistance is important not only clinically, but should also provide fundamental insights into the regulation of TSH secretion and the action of thyroid hormones.
