|
|
|
Indian Pediatr 2015;52:
489-492 |
 |
Energy and Protein Intake During Pregnancy in
Relation to Preterm Birth:
A Case Control Study
|
|
Shally Awasthi, M Chauhan, M Pandey, S Singh and *U
Singh
From Department of Pediatrics and *Obstetrics and
Gynecology, King George’s Medical University, Lucknow, UP, India.
Correspondence to: Dr Shally Awasthi, Professor,
Department of Pediatrics, King George‘s Medical University, Lucknow,
Uttar Pradesh, India.
Email: [email protected]
Received: August 02, 2014;
Initial review: September 08, 2014;
Accepted: March 10, 2015.
|
Objective: To find the association of maternal energy and protein
intake with preterm birth.
Design: Case-control study.
Setting: Two hospitals at
Lucknow in Northern India.
Participants: Cases (n=350)
were defined as mothers (age 18-40 y) of singleton live preterm (<37
wks) neonates. Controls (n=350) were mothers who delivered a
singleton neonate, consecutive to enrolled case, after completing 37
weeks of gestation.
Results: There was a
statistically significant lower mean (SD) energy intake [cases 1624
(249) Kcal vs. controls 1911 (341) Kcal; P<0.001] and
protein intake [cases 32.1 (6.1) vs. controls 37.2 (7.0); P<0.001]
among women who delivered preterm neonates. Maternal energy and protein
intake had significant positive correlation with neonatal weight,
length, foot length, head circumference and chest circumference.
Conclusion: Lower energy and
protein intake during pregnancy is possibly associated with preterm
birth.
Keywords: Diet, Low birth weight,
Prematurity.
|
|
Preterm birth is a major determinant of neonatal
mortality and childhood morbidity [1]
.
There seems to be an upward trend [2,3], which contributes to almost 3.6
million preterm births accounting for 23.6% of the global preterm births
reported each year [4]. Preterm birth has been consistently associated
with maternal underweight and nutrient deficiencies during pregnancy
[5-9]. Therefore, nutritional assessment needs to be an integral part of
antenatal care and should be continued during pregnancy in order to
reduce the risk of maternal, fetal and neonatal complications, as well
as the short- and long-term adverse outcomes. We conducted this
case-control study with the objective to find the association of
maternal energy and protein intake with preterm birth.
Methods
This was a hospital-based case-control study
conducted in King George’s Medical University, a tertiary-care teaching
institute, and Ram Manohar Lohia Hospital, a tertiary-care government
non-teaching institute, in Lucknow, Uttar Pradesh, India. These two
hospitals were chosen on the basis of their highest delivery data per
year. Ethical clearance for the study was obtained from the
Institutional ethical committee of King George’s Medical University.
Written informed consent was obtained from participating mothers.
The gestational age of neonate was assessed by using
modified Ballard score within 48 hours of birth [10]. Preterm was
defined as birth less than 37 weeks of gestation [1]. Cases were defined
as mothers (age 18-40 years) of live preterm neonates. Controls were
eligible mothers who delivered a neonate at or after 37 weeks of
gestation, consecutive to an enrolled case. Mothers who gave birth to
twins or those with neonates having congenital abnormalities were
excluded.
To detect a mean difference of 5 gm/day (SD 7 g) in
protein intake (two sided a=0.05,
power=90%), we required a sample of 292 women each for cases and
controls. We planned to recruit 350 cases and 350 controls.
Medical records were reviewed to obtain the
age, pregnancy details and illness status of included women. Data
were collected by a trained Medical scientist. Socioeconomic status was
defined using Modified Kuppuswamy‘s socioeconomic scale 2012 [11].
Social classes I and II were categorized as high, III as middle, and IV
and V as low. Mothers were asked for their daily intake of supplements
(Iron, folic acid, vitamin D and calcium) during pregnancy. A
predesigned questionnaire was used for obtaining the information of
their diet in one week preceding delivery. The questions were pertaining
to intake of milk, lentils, wheat products, rice, fruit vegetables, fast
food, meat products and Indian desserts. Food frequencies for individual
questions were converted into servings for a day or week, and then
summed to give a total daily (or weekly) intake of protein and energy.
Quantity was assessed by showing them household measurements like
katoris, teaspoon and glass of standard sizes. Nutritional
assessment was done on the basis of guidelines given by National
Institute of Nutrition [12].
All neonates were weighed on a weighing machine
corrected to one digit after decimal under the supervision of
pediatrician, within 48 hours of delivery. Neonatal length was measured
with infantometer. Neonatal foot length, head circumference and chest
circumference were measured within 48 hours by non-stretchable measuring
tape. Maternal Mid Upper Arm Circumference (MMUAC) was also obtained by
non-stretchable measuring tape.
Statistical Analysis: Data were analyzed by SPSS
version 14.0. Categorical data were compared by Chi-square test, and
Student’s t test was applied for continuous variables. Pearson’s
correlation coefficient was applied to analyze the correlation of
maternal diet with various anthropometric variables of newborn. P
value of <0.05 was considered to be significant.
Results
A total of 700 mothers (350 with preterm delivery and
350 with term delivery) from both hospitals (240 cases and controls each
from King George’s Medical University and 110 cases and controls each
from Ram Manohar Lohia Hospital) were recruited from April 2012 to April
2014 (Fig. 1).
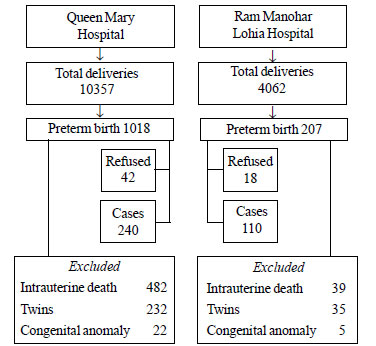 |
|
Fig.1 Flowchart showing recruitment of
cases.
|
Table I shows the sociodemographic and
anthropometric characteristics of cases and control mothers and their
newborns. Cases had lower Maternal Mid Upper Arm Circumference (MMUAC)
and lower post-delivery maternal weight.
TABLE I Socio-demographic and Anthropometric Characteristics of Cases and Controls
|
Characteristics
|
Case (n=350) |
Control (n=350) |
P value |
OR (95%CI)
|
|
Maternal |
|
age (y), mean |
26 (4.3) |
25.4 (3.8) |
0.07 |
1.0 (0.9-1.0) |
|
weight (Kg), mean (SD) |
52.1 (6.7) |
57.0 (5.9) |
<0.001 |
0.8 (0.8-0.9) |
|
Mid upper arm circumference (cm), mean (SD)
|
23.4 (2.3) |
24.4 (1.9) |
<0.001 |
0.8 (0.7-0.8) |
|
Vegetarian diet, n (%) |
210 (60.7%)
|
232 (66.3%)
|
0.12 |
0.7 (0.5-1.0) |
|
Illiterate, n (%) |
283 (80.9%) |
3 (0.9%) <0.001 |
488.5 |
(152.0-1570.0) |
|
Housewife |
334 (95.4%) |
320 (91.4%) |
0.04 |
1.9 (1.0 - 3.6) |
|
Socio Economic Status |
|
Middle (III) |
123 (25.7%) |
104 (30.2%) |
0.005 |
1.7 (1.1-2.5) |
|
Lower (IV and V) |
139 (39.4%) |
117 (33.9%) |
0.003 |
1.7 (1.2- 2.8)
|
|
Neonatal
|
|
Gestational age (wk), mean (SD) |
33.7 (1.9) |
38.4 (1.0) |
|
|
|
Weight (g), mean (SD) |
1850.8 (378.4) |
2820 (425.9) |
|
|
|
Whole length (cm), mean (SD) |
43 (2.4) |
46.9 (3.7) |
|
|
|
Foot length (cm), mean (SD) |
6.3 (0.6) |
7.6 (2.8) |
|
|
|
Head circumference (cm), mean (SD) |
30.3 (1.6) |
33.25 (1.1) |
|
|
|
Chest circumference (cm), mean (SD) |
28.0 (1.6) |
31.18 (1.1) |
|
|
TABLE II Comparison of Dietary Intake Between Cases and Controls
|
Diet |
Cases |
Controls |
P value |
|
Mean (SD) |
Mean (SD) |
|
|
Energy (Kcal/day)
|
1624 (249) |
1911 (341) |
<0.001 |
|
Protein (g/day) |
32 (6.0) |
37.2 (7.0) |
<0.001 |
|
Milk (mL/day) |
174 (66) |
175 (45) |
0.8 |
|
White clarified butter (g/day) |
2.2 (2.9) |
4.4 (3.6) |
<0.001 |
|
Lentils (g/day) |
67.6 (26.2) |
73.8 (28.5) |
0.003 |
|
Wheat (g/day) |
136.4 (29.5) |
147.1 (33.2) |
<0.001 |
|
Cooked rice (g/day) |
205.5 (87.3) |
241.5 (79.2) |
<0.001 |
|
Vegetables (g/day) |
284 (83.6) |
323.1 (93.3) |
<0.001 |
|
Fish (pieces/week) |
3.4 (1.1) |
2.0 (0.4) |
0.3 |
|
Chicken (pieces /week) |
2.4 (2.2) |
2.2 (0.6) |
0.8 |
|
Mutton (pieces /week) |
2 (0.7) |
1.8 (0.3) |
0.5 |
|
Dry fruits (no./week) |
5.4 (2.2) |
5.3 (1.7) |
0.5 |
|
Indian desserts (no./week) |
1.6 (0.5) |
1.8 (0.8) |
0.1 |
|
Fruits (no./week) |
1.5 (0.3) |
1.8 (0.4) |
0.9 |
Table II shows the dietary intake of mother
in the week preceding the delivery. Mothers who delivered neonates
before 37 weeks of gestation consumed lesser energy than the mothers who
delivered term neonates [1624 (240) Kcal vs. 1911 (341) Kcal; (P<0.001).
Mothers with term neonates consumed significantly more servings of
wheat, lentils, vegetables and fruits than mothers with preterm
neonates. Positive correlation was also observed between neonatal
anthropometric measures (head circumference, chest circumference,
neonatal whole length, foot length and weight), and maternal energy and
protein intake (Table III).
TABLE III Correlation of Neonatal Anthropometric Measurements with Maternal Dietary
Intake and Anthropometric Measurements
| |
Pearson Correlation Coefficient (P value) |
|
Neonatal
|
Neonatal
|
Foot
|
Head |
Chest |
Gestational
|
|
weight |
Whole length |
length |
circumference |
circumference |
age
|
|
Post delivery maternal weight |
0.3 (<0.001) |
0.2 (<0.001) |
0.1 (<0.001) |
0.3 (<0.001) |
0.3 (<0.001) |
0.3 (<0.001) |
|
Maternal mid upper arm circumference |
0.2 (<0.001) |
0.1 (<0.001) |
0.2 (<0.001) |
0.2 (<0.001) |
0.2 (<0.001) |
0.2 (<0.001) |
|
Maternal energy intake |
0.2 (<0.001) |
0.2 (<0.001) |
0.06 (0.07) |
0.3 (<0.001) |
0.4 (<0.001) |
0.4 (<0.001) |
|
Maternal protein intake
|
0.2 (<0.001) |
0.2 (<0.001) |
0.1 (0.01) |
0.3 (<0.001) |
0.4 (<0.001) |
0.3 (<0.001) |
Positive correlation was also observed between
maternal anthropometric measures and energy intake. Maternal daily
energy intake was positively associated with maternal weight (r=0.2,
P=0.001) and MMUAC (r=0.2, P<0.001). Protein intake was also
significantly correlated with maternal weight (r=0.2, P<0.001)
and MMUAC (r=0.1, P=0.002).
Discussion
In this case-control study, we observed a
statistically significant association of preterm birth with lesser
maternal energy and protein intake. Maternal energy and protein intake
also positively correlated with neonatal head circumference, chest
circumference, birth weight, whole length and foot length.
The limitation of this study was that information of
dietary intake in last one week was collected which is unlikely to
capture the nutritional intake during entire pregnancy. To overcome the
limitation of paucity of information on maternal diet, we also measured
Maternal Mid Upper Arm Circumference which is reported to be a potential
indicator of chronic nutritional status [13]. Recall method for
estimating dietary intake and approximation of quantity of food consumed
were the other limitations. The likely effect of other maternal,
placental and fetal factors leading to preterm birth was also not
evaluated in our study.
Our findings are supported by a Cochrane review that
concluded that less energy intake during pregnancy is strongly
associated with poor fetal growth, and underweight during pregnancy can
also be possibly associated with the risk of preterm birth [14]. On the
other hand, meta-analysis of five trials on balanced protein energy
supplementation during pregnancy showed no difference between the
incidence of term and preterm birth [15].
Our findings of positive correlation between maternal
energy and protein intake and neonatal anthropometric measures are
supported by another Indian study, reporting association of higher
energy intake during pregnancy with birth weight [16]. Walker, et al.
[17] reported that weight of the mother during pregnancy was associated
with higher head circumference, chest circumference and whole length of
neonate.
We conclude that lesser protein and energy intake is
possibly associated with preterm birth. Although etiology of preterm
birth is multi-factorial, maternal nutrition being a modifiable factor
can be used in public health interventions to prevent preterm
deliveries.
Contributors: All authors have contributed,
designed and approved the manuscript.
Funding: Indian Council of Medical Research
(5/7/513/10-RHN); Competing interests: None stated.
|
What is Already Known?
• Impaired language skills occur in children
with Phenylketonuria.
What This Study Adds?
• Early-treated and late-treated subjects
with PKU experience language impairment, but impairment is more
severe in late-treated subjects.
|
References
1. Preterm birth Fact sheet. N°363, WHO, Available
from: http://www.who.int/ media centre / factsheets/fs363/en.
Accessed November 28, 2014.
2. Piso B, Koss IZ, Winkler R. Antenatal
interventions to reduce preterm birth: An overview of Cochrane
systematic reviews. BMC Res Notes. 2014; 23: 265.
3. Howson EC, Kinney MV, Lawn JE. Born Too Soon: The
Global Action Report on Preterm Birth. World Health Organization.
Geneva, 2012.
4. Indian Foundation of Premature. Available from:
http://www.dayofbangalore.com/
news/healthcare/indian-foundation-premature-babies-ifpb-marks-world-prematurity.
Accessed November 28, 2014.
5. Bhattacharya S, Campbell DM, Liston WA,
Bhattacharya S. Effect of body mass index on pregnancy outcomes in
nulliparous women delivering singleton babies. BMC Public Health.
2007;7:168-73.
6. Dietz PM, Callaghan WM, Cogswell ME, Morrow B,
Ferre C, Schieve LA. Combined effect of pre-pregnancy body mass index
and weight gain during pregnancy on the risk of preterm delivery.
Epidemiology. 2006;17:170-7.
7. Cnattingius S, Bergstrom R, Lipworth L, Kramer MS.
Pre-pregnancy weight and the risk of adverse pregnancy outcomes. N Engl
J Med. 1998;338:147-52.
8. McDonald SD, Han Z, Mulla S, Beyene J; Knowledge
Synthesis Group. Overweight and obesity in mothers and risk of preterm
birth weight infants: Systematic review and meta-analysis. BMJ.
2010;341:c3428.
9. Xinxo S, Bimbashi A, Kakarriqi EZ, Zaimi E.
Association between maternal nutritional status of pre pregnancy,
gestational weight gain and preterm birth. Mater Sociomed. 2013;25:6-8.
10. Ballard JL, Khoury JC, Wedig K, Wang L, Eilers-Walsman
BL, Lipp R. New Ballard Score, expanded to include extremely
premature infants. J Pediatr. 1991;119:417-23.
11. Bairwa M, Rajput M, Sachdeva M. Modified
Kuppuswamy’s socioeconomic scale: Social researcher should include
updated income criteria, 2012. Indian J Community Med. 2013:38:185-6.
12. Dietary Guidelines for Indians. National
Institute of Nutrition. 2010. Available From:
http://ninindia.org/DietaryguidelinesforIndians-Finaldraft.pdf.
Accessed November 28, 2014.
13. Ricalde AE, Velasquez-Melendez G, Tanaka AC, De
siqueria AA. Mid upper arm circumference in pregnant women and its
relation to birth weight. Reu Saude Publica. 1998:112-7.
14. Kramer MS, Kakuma R. Energy and protein intake in
pregnancy. Cochrane Databse Syst Rev. 2003;4:CD000032.
15. Villar J, Merialdi M, Gulmezoglu AM, Abalos E,
Carroli G, Kulie R, et al. Nutritional interventions during
pregnancy for the prevention or treatment of maternal morbidity and
preterm delivery: An overview of randomized controlled trials. J Nutr.
2003;133:1606-25.
16. Bhatia BD, Banerjee D, Agarwal DK, Agarwal K.
Fetal growth: Relationship with maternal dietary intakes. Indian J
Pediatr. 1983;50:113-20.
17. Walker SP, Ewan-Whyte C, Chang SN, Powell CA,
Feltcher H, Donald DM, et al. Factors association with size and
proportionality at birth in term Jamaican infants. J Health Popul Nutr.
2003;21:117-26.
|
|
|
 |
|

