Postnatal growth of very low
birth weight (VLBW) neonates has always remained a challenge in NICU.
They are often less than the 10
th
percentile of reference intrauterine curves at the time of hospital
discharge [1]. The AAP Committee on Nutrition recommendation of
providing nutrient intakes that permit the rate of postnatal growth to
approximate that of a normal fetus of the same postmenstrual age is
rarely met for very preterm babies [2].
Early aggressive nutritional management as a solution
to postnatal growth failure has been studied. The evidence supports
recommendations to administer early parenteral nutrition and enteral
nutrition [1]. There is evidence from randomized controlled trials that,
early aminoacid supplementation (starting within few hours of birth) as
compared to amino acid supplementation after 3-5 days of life, in very
preterm babies, results in better post natal growth [3,4]. Previous
studies [5-9] have shown that higher doses of amino acids from day 1
result in better plasma amino acid profile and nitrogen accretion and
are well tolerated, but its role in improving postnatal growth rate and
long term neurodevelopment is still inconclusive.
In resource limited settings, use of lipids and
micronutrients is restricted for various reasons, and partial parenteral
nutrition (comprising of glucose, amino acids and electrolytes, but no
lipids) is administered to the preterm babies. We undertook this trial
to assess if a better post natal growth could be demonstrated in VLBW
infants by starting parenteral amino acids at a higher dose (3 g/kg/d)
from day 1 of life in comparison to gradual increments in dose of
parenteral amino acids in the partial parenteral nutrition regimen.
Methods
This single centre randomized controlled trial
enrolled inborn babies with birth weight 900-1250 g from a level 3 NICU
in Mumbai between February 2008 and February 2010. Babies missed out in
the first 24 hrs of life, having obvious congenital anomalies affecting
growth and requiring surgical intervention were excluded from the study.
A written informed consent was obtained from the parents before
enrolment. The study was approved by the Institutional ethics committee.
The subjects were randomized into two groups within
first 24 hours of life to receive 2 different doses of parenteral amino
acid preparation. The Partial Parenteral Nutrition (PPN) administered to
the infant in both groups was composed of dextrose, amino acids, sodium
and potassium. Lipids, multivitamin and trace elements were not
routinely provided. The PPN was administered through peripheral
intravenous line. The Low AA group received 1 g/kg/d of parenteral amino
acids on day 1 and dose increased by 1 g/kg every day till maximum of 4
g/kg/d. The High AA group received 3 g/kg/d of parenteral amino acids on
day 1 and dose increased to 4 g/kg/d on next day.
Sample size calculations: In a pilot observation
in our unit, amongst babies with weight between 900 to 1250 grams,
average weight gain of a baby at the end of day 28 of life, who receives
parenteral amino acid (PPN) similar to those in Low AA group was found
to be 10 grams/kg/day. We hypothesized that, by starting amino acid
supplementation at 3g/kg/d from day 1 of life, as in High AA group,
weight gain would improve at least by 50% (that is 15 g/kg/d). Assuming
SD of 10%, a sample size of 63 babies would be required in each group
for a 2-tailed alpha error of 0.05 and beta error of 0.20 (power 80%).
We enrolled 75 babies in each group to account for drop out due to death
or loss of follow up within 28 days of life.
Randomization: A random number sequence
was generated in a variable block size of two or four each using a
"Random Allocation Software" computer program. The random allocation of
sequence was generated by a statistician who was not a part of the
study. The random codes were kept in serially numbered, opaque, sealed
and identical envelopes to eliminate selection bias. The participants
were enrolled by the on call senior residents working in the NICU.
Eligible infants were assigned to interventions by them by opening
sequentially numbered sealed opaque envelopes.
Blinding: The composition of the study solutions
for seven different weight groups of 50 grams each between 900 and 1250
g and for the first 8 days of life (time to reach maximum fluid volume
in each group) was provided by the investigator in two tabular sheets
(for 2 study groups) to the staff nurse responsible for preparing PPN.
This nurse was not involved in patient care. The senior resident who
assigned the babies to the intervention was not involved in the study.
Thus, blinding was achieved at two levels (clinicians involved in care
of the infant and judicial assessors of outcomes).
Primary outcome was postnatal growth at 28 days of
age as defined by weight gain by 28 days in g/kg/day. Weight gain by 28
days was calculated as (weight in grams at 28 days - birth wt in
grams)/birth wt in kg/28. Secondary outcomes were 1) weight (g), length
(cm), and head circumference (cm) at 28 days 2) gain in length and head
circumference by 28 days (cm/wk) 3) number of days on PPN 4) the number
of days required to regain birth weight 5) duration of hospital stay and
6) the incidence of following morbidities: patent ductus arteriosus
(PDA), sepsis (early and late onset), intraventricular hemorrhage (IVH),
necrotizing enterocolitis (NEC), chronic lung disease (CLD/BPD),
retinopathy of prematurity (ROP), hypoglycemia and anemia in both
groups.
Babies were weighed on an electronic weighing scale
(accuracy of 5 g) immediately after birth and subsequently daily till
discharge. Length and head circumference were measured by investigator
at birth and subsequently, weekly till discharge. Weight, length and
head circumference were measured on follow-up at 28 days of age.
Incidence of PDA, sepsis, necrotizing enterocolitis (NEC),
intraventricular hemorrhage (IVH), chronic lung disease (CLD),
retinopathy of prematurity (ROP), hypoglycemia were assessed in both
groups. PDA was diagnosed by the presence of continuous murmur in the
left 2
nd intercostal space
and one of the following- tachycardia, bounding peripheral pulses, pulse
pressure of >25 mm of Hg, hyperkinetic precordium, and hepatomegaly.
Sepsis was diagnosed with either a positive body fluid culture or
nephalometric CRP>10mg/l (early and late onset sepsis were defined based
on the onset of sepsis before or after 72 hrs of life). Ultrasound
grading of IVH was performed by grading system described by Papile [10].
NEC was diagnosed by the presence of clinical triad of abdominal
distension with thrombocytopenia and hyponatrimia or suggestive
radiological findings (pneumatosis intestinalis, pneumoperitoneum).
CLD/BPD was defined by the need of supplemental oxygen at 28 days of
life [11]. Stages of ROP were diagnosed as per the international
classification (ICROP) [12]. Anemia was defined by hemoglobin value of
<13g/dL (assessed within 4 weeks of life) and hypoglycemia by a blood
sugar of <40mg/dL.
For babies weighing <1 kg and
³1 kg, total fluid
intake on day 1 was 100 mL/kg/d and 80 mL/kg/d, respectively. Additional
20 mL/kg/d were provided in both groups from day 1 for extra insensible
water losses due to radiant warmer. Total fluids were increased by 20 mL/kg
every day in both groups till a maximum of 190 mL/kg/d and 170 mL/kg/d
by day 8 in babies weighing <1kg and
³1kg respectively.
Fluids were administered in both the groups such that the glucose
infusion rate was not less than 5 mg/kg/min and non protein calorie
intake not less than 35 Kcal/kg/d. Both groups received parenteral 10%
dextrose, parenteral aminoacid preparation and electrolytes (after day 3
of life). In this study, parenteral lipids were administered only to the
babies weighing <1 kg beginning at the rate of 0.5 g/kg/d and advancing
0.5 g/kg/d to a maximum of 3.5 g/kg/d. Lipids were not routinely
administered in the study group, as per unit policy.
All babies in the study were started on trophic feeds
(10 mL/kg/d) on day 1 and feeds were not advanced for the first 4 days
(till the dose advancement of parenteral amino acid supplements was
complete). Subsequently feeds were advanced at the rate of 10-15 mL/kg/d
if babies tolerated feeds and were hemodynamically stable. As enteral
feeds were advanced, intravenous administration of PPN study solutions
were decreased accordingly keeping the total fluid requirement of the
baby constant as per standard practice. Feeds were predominantly
expressed breast milk (EBM) / Pasteurized donor milk from Human Milk
Bank. Formula feeding is not allowed in our unit. After achieving full
enteral feeds, fortification was indicated for babies with moderate and
severe CLD, PDA with congestive cardiac failure, where poor weight gain
was due to restricted feed volumes of <150 mL/kg/d, which constituted
<10% babies in this study. Human milk fortification was discontinued at
discharge.
All babies in the study received parenteral amino
acids till enteral feeds constituted 75% of the total fluid requirement
of the baby
Statistical analysis: All data were analysed
using SPSS version 16 statistical package. Continuous variables with
normal distribution (gestational age, weight at birth and 28 days,
length at birth and 28 days, weight and length gain) were analyzed by
two sample t test. Head circumference at birth and 28 days were
not normally distributed and hence analysed by Mann Whitney U test.
Kolmogorov Smirnov test was used to check normality of continuous data.
Categorical data was assessed using Fischer Exact test. A P value
of <0.05 was considered as statistically significant. Analysis was
performed by following intention to treat principle.
Results
Between February 2008, and February 2010, 150
neonates were enrolled (Fig. 1). Out of the 150 patients
enrolled, 123 completed the study. There were no significant differences
in the mean birthweight, gestational age, APGAR scores, number of SGA
babies between two treatment groups (Table I).The baseline
characteristics in the neonates that could not be followed up at 28 days
of life, were similar in both the groups. Only 16 out of 150 babies
received parenteral lipid preparations. There was no difference in the
number of days of parenteral amino acid support.
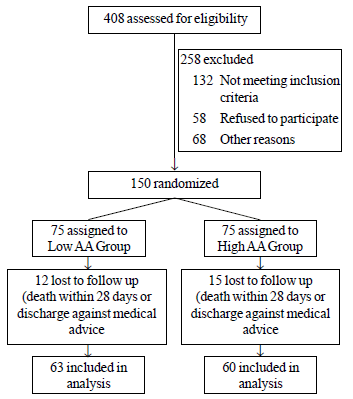 |
|
Fig. 1 Flow diagram of patients in
study.
|
TABLE I Baseline Characteristics of Infants
|
Baseline Characteristics |
Low AA Group
(n=75) |
High AA
group (n=75) |
|
Gestational age (wks) * |
32.12 (2.3) |
31.65 ± 1.97 |
|
Birthweight (g)* |
1092.4 (105.5) |
1103.5 ± 110.1 |
|
Length (cm)* |
37.63 (2.65) |
37.94 ± 2.17 |
|
Head circumference † (cm) |
26 (26-28) |
27 (27-28) |
|
1 min APGAR <5
|
15 (20) |
12 (16) |
|
5 min APGAR<5
|
6 (8) |
5 (6.6) |
|
Male gender
|
35 (46.6) |
26 (34.6) |
|
SGA |
30 (40) |
24 (32) |
|
Multiple pregnancy
|
7 (9.3) |
5 (6.6) |
|
Antenatal steroid use
|
10 (13.3) |
14 (18.6) |
|
Mechanical ventilation
|
4 (5.3) |
8 (10.6) |
|
CPAP
|
4 (5.3) |
12 (16) |
|
Surfactant
|
3 (4) |
6 (8) |
|
Inotropic support
|
8 (10.6) |
11 (14.6) |
|
Parenteral lipids
|
7 (9.3) |
9 (12) |
|
Fluid restriction
|
18 (24) |
10 (13.3) |
|
Feed fortification
|
8 (10.6) |
6 (8) |
|
Postnatal steroid use
|
4 (5.3) |
5 (6.6) |
|
* Data represented as Mean (± SD), and
†median (IQR); Rest in number (%); CPAP: Continous positive air
way pressure. |
TABLE II Postnatal Growth and Nutritional Intake Among The Study Subjects
|
Outcome |
Low AA Group
|
High AA Group
|
Mean difference
|
P value |
|
(n=63) |
(n=60) |
(95% CI) |
|
|
Weight gain at 28 days (g/kg/d)* |
13.15 (5.25) |
8.67 ± 4.28 |
4.48 (2.76-6.19) |
<0.001
|
|
Weight in g at 28 days* |
1494.7 (224.4) |
1371.58 ± 202.64 |
123.12(46.67-199.57) |
0.01
|
|
Length in cm at 28 days* |
40.21 (2.34) |
39.19 ± 1.8 |
1.02(0.27-1.77) |
0.008
|
|
Length gain (cm/wk)* |
0.63 (0.36) |
0.36 ± 0.348 |
0.27(0.14-0.39) |
<0.001
|
|
Head circumference in cm at 28 days† |
29 (27.5-30.5) |
28 (27-29) |
|
0.42
|
|
Head circumference gain (cm/wk)† |
0.625 (0.37-0.875) |
0.25 (0.03-0.59) |
|
<0.001
|
|
Total days of PPN† |
12 (7-15) |
10 (7-15) |
|
0.31
|
|
Cumulative Enteral intake in first 28 days (kcal/kg/d)* |
69.9 (3.99) |
71.1 ± 3.34 |
-1.2(-2.52-0.11) |
0.07
|
|
Cumulative non protein calorie from PPN (kcal)* |
349.1 (55.34) |
318.5 ± 60.43 |
30.6(9.93-51.27) |
0.004
|
|
Time to regain BW (days)† |
12 (10-14) |
16 (11-20) |
|
<0.001
|
|
Duration of hospital stay (days)† |
21 (14-26) |
19 (13-26) |
|
0.25
|
|
*Data represented as mean (SD); †Data represented as median
(IQR). |
The primary outcome of weight gain by 28 days was
13.15 g/kg/d and 8.67 g/kg/d in Low AA group and High AA group,
respectively (P<0.001) (Table II).The weight and
length at 28 days of life were higher in the Low AA group (Fig.
2). Median gain in head circumference was higher in the Low AA
group. After adjusting for important baseline clinical variables that
may act as confounders (multiple pregnancy, antenatal steroids, 5 minute
APGAR score, birth weight, gender, SGA status, early onset sepsis, late
onset sepsis, respiratory support, inotropic support and hypo-glycemia),
using multivariate linear regression analysis, High AA group had
significantly lower rate of weight gain (g/kg/day) compared to the Low
AA group. No other variables were significantly associated with the rate
of weight gain.
We did not find significant differences in total
duration of PPN, duration of hospital stay, frequency of PDA, sepsis,
IVH, NEC, hypoglycemia, anemia, CLD and ROP between the two groups (Table
III).
TABLE III Neonatal Morbidity in The Two Groups No.(%)
|
Low AA
|
High AA
|
|
Group (n=63) |
Group (n=60) |
|
Patent ductus arteriosus |
16 (25.4) |
7 (11.6) |
|
Early onset sepsis |
28 (44.4) |
19 (31.6) |
|
Late onset sepsis |
9 (14.3) |
7 (11.6) |
|
Intraventricular hemorrhage |
5 (7.9) |
6 (10) |
|
Necrotising enterocolitis |
6 (9.5) |
8 (13.3) |
|
Chronic lung disease |
6 (9.5) |
6 (10) |
|
Retinopathy of prematurity |
2 (3.2) |
3 (5) |
|
Anemia |
8 (12.7) |
6 (10) |
|
Hypoglycemia |
10 (15.8) |
16 (26.6) |
|
P value >0.05 for all comparisons. |
Discussion
We found that 1g/kg/d of parenteral amino acids on
day 1 with gradual increments of 1g/kg every day till a maximum of
4g/kg/d resulted in better growth (weight, length and head
circumference) than early aggressive parenteral amino acid
supplementation (3g/kg on day 1 of life) in very low birth weight
infants. There were no significant differences in total duration of PPN,
duration of hospital stay, enteral intake and morbidities associated
with prematurity, between the two groups.
Strengths of our study were: robust randomized
controlled trial design, sufficient sample size with adequate power to
detect a difference. The blinding was achieved at two levels: clinicians
involved in care of the infant and assessors of outcomes. Limitations of
our study include: partial parenteral nutrition with inadequate
calories, short term assessment of postnatal growth, and absence of
biochemical evidence of protein accretion to support clinical evidence.
Randomized controlled trials [5-9] on aggressive
protein supplementation have reported no difference in the weight gain
in the high versus low AA group. However, majority of those [5-8] were
designed to study amino acid profile and nitrogen retention, and were
not powered to study the outcome of postnatal growth.
The mean gestational age in most other studies on
aggressive protein supplementation was <30wks and the mean birthweight
was between 800-950g [3-7, 9]. Most of the studies included ventilated
babies only. Clark, et al. [9] reported surfactant use in >75% of
the babies, whereas only 18% of our babies needed any form of
respiratory support. In other words, babies in our study groups were
larger, more mature and required less respiratory support than in most
of the trials on aggressive protein supplementation, yet there was poor
weight gain in the High AA group.
High AA group had more babies on respiratory support
than the low AA group; and 50% of the babies in the study had sepsis.
However, the regression analysis showed that the weight difference
between the two study groups was influenced only by the dose of amino
acids. The cumulative enteral calorie intake, time to reach full feeds
and the number of babies on fortified feeds were also similar between
the groups suggesting that the nutrition intake in the later part of the
study period (after the 1
st
week) was similar. Therefore, the poor weight gain in high AA group was
attributed to the lower non protein calorie supplementation through PPN
in the first week of life (more marked in the first three days) of life,
resulting in a low calorie nitrogen ratio (CNR) - which occurred because
of aggressive protein supplementation in the settings of partial
parenteral nutrition
A CNR (ratio of non protein calories: calories from
protein) of 150-250 through parenteral nutrition, is desired for post
natal growth. [13]. A minimum non protein calorie intake of at least
35-40 kcal/kg/d is required for the utilization of 1g/kg of protein
supplemented per day. For higher doses of protein, the non protein
calorie intake should also increase proportionately to optimise the CNR
and improve postnatal growth. [14]. Most of the studies involving higher
doses of amino acids also administered parenteral lipids in both groups
from day 1 [5-9]. In current study, 90% of the babies received partial
parenteral nutrition (i.e. without lipid infusion). This
reduction in non-protein calorie intake, along with higher doses of
amino acids, caused more than two fold reduction in the CNR in the high
AA group in the first three days of life (The non-protein energy intake
in the Low AA group and High AA group was 44 kcal/kg and 36 Kcal/kg,
respectively on day 1 of life resulting in the calorie nitrogen ratio of
150 in Low AA group and 62.5 in High AA group). After day 4 of life, the
advancement of parenteral protein was complete; the enteral intake was
increased and the volume of dextrose in the PPN increased to meet the
daily fluid requirements. This improved the non-protein calorie intake
(56-70 Kcal/kg/d) -though still inadequate in both the groups. After the
1
st week of life, increments
in the enteral nutrition optimized the non-protein calorie intake.
Essentially after day 4, the protein and non-protein calorie intake
provided through the study solutions were the same in both groups.
The best option to optimize the CNR in the high AA
group would have been the provision of lipids in both groups from day 1
of life. It was not feasible to administer lipids to all babies in this
study. The next option would have been provision of higher
concentrations of dextrose in the study solutions. As we used 10%
dextrose and amino acids in variable proportions to prepare the study
solutions, the net dextrose concentration in the High AA solution varied
from 7-8%. We did not attempt to achieve the final concentration of
dextrose at 10% in PPN fluid in either group for following reason. In
past, in our unit, addition of higher concentrations of dextrose to
obtain a 10% dextrose study solution resulted in a higher incidence of
hyperglycemia in the ELBW babies in the first week of life, especially
with infection.
The delay in regaining birth weight in the High AA
group could be attributed to the inadequate calorie intake in the High
AA group in the first week of life. The mean duration of PPN in our
study was lower than that reported in the NICHD Neonatal Research
Network study [15]. Interestingly, the babies in the Low AA group did
not get discharged earlier than the High AA group, despite a better
weight gain, because they had to be nursed till the attainment of
full-suck feeds. The duration of hospital stay for the babies in this
study was much lower, because we could follow them up in our Kangaroo
Mother Care Clinic after discharge, on a daily basis.
This study also highlights the fact that increments
in weight, length and head circumference in the study groups were less
than those described in the studies done in the Western world. This
explains the need for optimizing calorie intake soon after birth, as
well as for long term data on growth pattern and neurodevelop-mental
outcome in these infants.The consistent finding in most studies, that
weight gain was unaffected by differing protein intake, could be
attributed to the similar calorie intake between the groups, triggering
a debate on the importance of adequate calorie supplementation over
protein for preterm babies
This study underscores the need of research in the
role of PPN in babies <1000g, that require parenteral nutrition for
longer duration. It is also mandatory to know if PPN affected long term
growth in the very preterm babies, bearing in mind the implications of
rapid catch up growth in the preterm babies with poor weight gain in the
first month of life. Also, the long term neurodevelopment in preterm
babies that are deprived of the essential fatty acids as a result of PPN
in the first week of life, needs to be studied
We conclude that, starting higher doses of amino
acids without parenteral lipids and higher dextrose concentrations did
not result in a better weight gain than slow dose advancement.
Therefore, in resource limited settings, where parenteral lipids are not
started within first 3 days of post-natal life, starting amino acids at
1g/kg/d on day 1 and gradual advancement would be a better option.
Acknowledgments: The authors thank the Dean, Seth
GS Medical College and KEM Hospital, Mumbai for permitting them to
publish the manuscript. The authors also acknowledge Dr. Shripada C Rao
(Prof and Head, Department of Neonatology, Princess Mararet Hospital for
Children, Western Australia for help in statistical analysis.
Contributors: HB: was involved in the review of
literature, data collection and wrote the draft of the paper; RNN: was
involved in management of patients, designing of study and drafting the
manuscript and will act as a guarantor; NSK: was involved in drafting
the article, analysis and interpretation of data. The final manuscript
was approved by all the authors.
Funding: None; Competing interests: None
stated.
|
What is Already Known?
• Early aggressive parenteral protein
supplementation in TPN in preterm neonates improves rate of
protein accretion, but not post natal growth.
What This Study Adds?
• In resource limited settings, where partial
parenteral nutrition is provided, an initial dose of 1g/kg/d of
parenteral protein followed by gradual advancement to 4g/kg/d
results in better postnatal growth than aggressive protein
supplementation.
|
References1. Ehrenkranz RA. Early, aggressive nutritional
management for very low birth weight infants: what is the evidence?
Semin Perinatol. 2007;31:48-55.
2. Ziegler EE, Thureen PJ, Carlson SJ. Aggressive
nutrition of the very low birth weight infant. Clin Perinatol.
2002;29:225-44.
3. Poindexter BB, Ehrenkranz RA, Stoll BJ, Koch MA,
Wright LL, Oh W. Effect of parenteral glutamine supplementation on
plasma amino acid concentrations in extremely low-birth weight infants.
Am J Clin Nutr. 2003;77:737-43.
4. Valentine CJ, Fernandez S, Rogers LK, Gulati P,
Hayes J, Lore P, et al. Early amino-acid administration improves
preterm infant weight. J Perinatol.2009;29:428-32.
5. Ibrahim HM, Jeroudi MA, Baier RJ, Dhanireddy R,
Krouskop RW. Aggressive early total parenteral nutrition in low birth
weight infants. J Perinatol. 2004;24:482-6.
6. Thureen PJ, Melara D, Fennessey PV, Hay WW Jr.
Effect of low versus high intravenous amino acid intake on very low
birth weight infants in the early neonatal period. Pediatr Res.
2003;53:24-32.
7. Blanco CL, Gong AK, Green BK, Falck A, Schoolfield
J, Liechty EA. Early changes in plasma amino acid concentrations during
aggressive nutritional therapy in extremely low birth weight infants. J
Pediatr. 2011;158:543-8.
8. Bulbul A, Okan F, Bulbul L, Nuhoglu A. Effect of
low versus high early parenteral nutrition on plasma amino acid profiles
in very low birth- weight infants. J Matern Fetal Neonatal Med.
2012;25:770-6.
9. Clark RH, Chace DH, Spitzer AR, Pediatrix Amino
Acid Study Group. Effects of two different doses of amino acid
supplementation on growth and blood amino acid levels in premature
neonates admitted to the neonatal intensive care unit: a randomized,
controlled trial. Pediatrics. 2007;120:1286-96.
10. Papile LA, Burstein J, Burstein R, Koffler H.
Incidence and evolution of subependymal and intraventricular hemorrhage:
a study of infants with birth weights less than 1,500 gm. J Pediatr.
1978;92:529-34.
11. Bancalari E. Bronchopulmonary dysplasia. Semin
Neonatol. 2003;8:63-71.
12. An international classification of retinopathy of
prematurity. Pediatrics. 1984;74:127-33.
13. Kalhan SC, Price PT. Nutrition and selected
disorders of the gastrointestinal tract. In: Klaus MH, Fanaroff
AA, editors. Care of High risk Neonate. 5th ed. Philadelphia: WB
Saunders; 2001. p. 147-94.
14. Kashyap S, Schulze KF, Ramakrishna R, Dell RB,
Heird WC. Evaluation of a mathematical model for predicting the
relationship between protein and energy intakes of low-birth-weight
infants and the rate and composition of weight gain. Pediatric Res.
1994;35:704-12.
15. Ehrenkranz RA, Younes N, Lemons JA, Fanaroff AA, Donovan EF,
Wright LL, et al. Longitudinal growth of hospitalized very low
birth weight infants. Pediatrics. 1999;104:280-9.

